This is a preprint.
Performance and usability evaluation of three LDH-based malaria rapid diagnostic tests in Kédougou, Senegal
- PMID: 39711730
- PMCID: PMC11661343
- DOI: 10.1101/2024.12.12.24318945
Performance and usability evaluation of three LDH-based malaria rapid diagnostic tests in Kédougou, Senegal
Update in
-
Performance and usability evaluation of three LDH-based malaria rapid diagnostic tests in Kédougou, Senegal.Parasit Vectors. 2025 Jul 12;18(1):280. doi: 10.1186/s13071-025-06914-9. Parasit Vectors. 2025. PMID: 40652282 Free PMC article.
Abstract
Background: The emergence of pfhrp2/3-deleted parasites threatens histidine-rich protein 2 (HRP2)-based malaria rapid diagnostic test (RDT) performance. RDTs targeting Plasmodium falciparum (Pf) lactate dehydrogenase (LDH) may address current product limitations and improve case management.
Objectives: To evaluate the performance and usability of three LDH-based RDTs in febrile patients.
Methods: A cross-sectional diagnostic accuracy study was conducted in Kédougou, Senegal. Capillary blood was tested using the SD Bioline Ag Pf (#05FK50) and three LDH-based RDTs: BIOCREDIT Malaria Ag Pf (pLDH), BIOCREDIT Pf (pLDH/HRPII), and BIOCREDIT Pf/Pv (pLDH/pLDH) (Rapigen Inc., Republic of Korea). Venous blood was collected to repeat the BIOCREDIT RDTs and conduct microscopy. Frozen venous specimens were tested using a reference PCR assay. A quantitative multiplex malaria antigen assay measured antigen concentration. RDT performance was determined and analyzed as a function of antigen concentration distribution. Usability of the Pf-only BIOCREDIT tests was evaluated using a questionnaire.
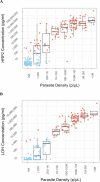
Results: 154/220 participants (70%) were Pf-positive by PCR. The Pf (pLDH/HRPII) test demonstrated the highest sensitivity at 78% (70.9%-84.5%); specificity was 89% (79.4%-95.6%). All RDTs performed better than microscopy (53% sensitivity). RDTs performed better when compared to antigen concentration over PCR results. Improved sensitivity of the Pf (pLDH/HRPII) test was driven by the HRP2 line. Line intensity correlated with antigen concentration. Predicted sensitivity using the analytical limit of detection (LOD) was comparable to observed sensitivity. RDTs demonstrated acceptable usability.
Conclusions: Both HRP2 and LDH contributed to the sensitivity of the best-performing Pf-RDT. RDT analytical LODs can be used to predict performance in populations with known antigen concentrations.
Keywords: Malaria; diagnosis; histidine rich protein 2; lactate dehydrogenase; rapid diagnostic test.
Conflict of interest statement
Conflict of interest statement The authors have no competing interests to declare.
Figures


References
-
- World Health Organization, Global Malaria Programme. How malaria RDTs work [Internet]; n.d. [Cited 2024 Feb 19]. Available from: https://www.who.int/teams/global-malaria-programme/case-management/diagn...
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
