Conformational Flexibility of D1-Glu189: A Crucial Determinant in Substrate Water Selection, Positioning, and Stabilization within the Oxygen-Evolving Complex of Photosystem II
- PMID: 39713658
- PMCID: PMC11656237
- DOI: 10.1021/acsomega.4c09981
Conformational Flexibility of D1-Glu189: A Crucial Determinant in Substrate Water Selection, Positioning, and Stabilization within the Oxygen-Evolving Complex of Photosystem II
Abstract
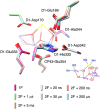
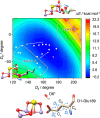
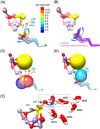
Photosynthetic water oxidation is a vital process responsible for producing dioxygen and supplying the energy necessary to sustain life on Earth. This fundamental reaction is catalyzed by the oxygen-evolving complex (OEC) of photosystem II, which houses the Mn4CaO5 cluster as its catalytic core. In this study, we specifically focus on the D1-Glu189 amino acid residue, which serves as a direct ligand to the Mn4CaO5 cluster. Our primary goal is to explore, using density functional theory (DFT), how the conformational flexibility of the D1-Glu189 side chain influences crucial catalytic processes, particularly the selection, positioning, and stabilization of a substrate water molecule within the OEC. Our investigation is based on a hypothesis put forth by Li et al. (Nature, 2024, 626, 670), which suggests that during the transition from the S2 to S3 state, a specific water molecule temporarily coordinating with the Ca ion, referred to as O6*, may exist as a hydroxide ion (OH-). Our results demonstrate a key mechanism by which the detachment of the D1-Glu189 carboxylate group from its coordination with the Ca ion allows the creation of a specialized microenvironment within the OEC that enables the selective attraction of O6* in its deprotonated form (OH-) and stabilizes it at the catalytic metal (MnD) site. Our findings indicate that D1-Glu189 is not only a structural ligand for the Ca ion but may also play an active and dynamic role in the catalytic process, positioning O6* optimally for its subsequent participation in the oxidation sequence during the water-splitting cycle.
© 2024 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

Similar articles
-
Oxygen-evolving photosystem II structures during S1-S2-S3 transitions.Nature. 2024 Feb;626(7999):670-677. doi: 10.1038/s41586-023-06987-5. Epub 2024 Jan 31. Nature. 2024. PMID: 38297122 Free PMC article.
-
Evidence from FTIR difference spectroscopy that D1-Asp61 influences the water reactions of the oxygen-evolving Mn4CaO5 cluster of photosystem II.Biochemistry. 2014 May 13;53(18):2941-55. doi: 10.1021/bi500309f. Epub 2014 Apr 23. Biochemistry. 2014. PMID: 24730551
-
The O2-Evolving Complex of Photosystem II: Recent Insights from Quantum Mechanics/Molecular Mechanics (QM/MM), Extended X-ray Absorption Fine Structure (EXAFS), and Femtosecond X-ray Crystallography Data.Acc Chem Res. 2017 Jan 17;50(1):41-48. doi: 10.1021/acs.accounts.6b00405. Epub 2016 Dec 21. Acc Chem Res. 2017. PMID: 28001034 Review.
-
Roles of D1-Glu189 and D1-Glu329 in O2 Formation by the Water-Splitting Mn4Ca Cluster in Photosystem II.Biochemistry. 2020 Oct 13;59(40):3902-3917. doi: 10.1021/acs.biochem.0c00541. Epub 2020 Sep 29. Biochemistry. 2020. PMID: 32931699
-
Biological water oxidation.Acc Chem Res. 2013 Jul 16;46(7):1588-96. doi: 10.1021/ar3003249. Epub 2013 Mar 18. Acc Chem Res. 2013. PMID: 23506074 Review.
Cited by
-
Probing substrate water access through the O1 channel of Photosystem II by single site mutations and membrane inlet mass spectrometry.Photosynth Res. 2025 Apr 22;163(3):28. doi: 10.1007/s11120-025-01147-4. Photosynth Res. 2025. PMID: 40263146 Free PMC article.
References
-
- Photosystem II: The Light Driven Water: Plastoquinone Oxidoreductase; Wydrzynski T. J.; Satoh K., Eds.; Springer: Dordrecht, The Netherlands, 2005.
LinkOut - more resources
Full Text Sources
