Nano hydroxyapatite-silica with a core-shell structure for long-term management of dentin hypersensitivity
- PMID: 39717093
- PMCID: PMC11664142
- DOI: 10.1016/j.isci.2024.111474
Nano hydroxyapatite-silica with a core-shell structure for long-term management of dentin hypersensitivity
Abstract
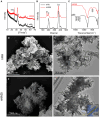
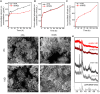
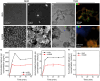
Teeth undergo continuous demineralization and remineralization influenced by dietary acid and saliva. Excessive dietary acid attack disrupts this balance, exposing dentin tubules and causing dental hypersensitivity (DH). Due to low acid resistance, traditional anti-DH regents such as calcium phosphate minerals fail in long-term occlusion of dentin tubules, resulting in recurrent attacks of DH. Hence, we fabricate nano hydroxyapatite (nHA)-silica (nHASi) with a core-shell structure that can not only fill in the dentin tubules, releasing Ca2+ and PO4 3- from the nHA core for biomineralization, but also exhibit remarkable acid resistance due to the silica shell. Our study demonstrates a continuous growth of hydroxyapatite (HA) nanocrystals within nHASi during cyclic de/remineralization. When applied with toothpaste, nHASi gradually enhances dentin tubule occlusion over de/remineralization cycles. Additionally, extracts of nHASi exhibit excellent cytocompatibility and odontogenic inductivity in vitro. This work provides a paradigm for developing effective anti-allergic materials for the long-term management of DH.
Keywords: Applied sciences; Health sciences; Materials science.
© 2024 The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




References
-
- Degli Esposti L., Ionescu A.C., Brambilla E., Tampieri A., Iafisco M. Characterization of a Toothpaste Containing Bioactive Hydroxyapatites and In Vitro Evaluation of Its Efficacy to Remineralize Enamel and to Occlude Dentinal Tubules. Materials. 2020;13:2928. doi: 10.3390/ma13132928. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

