Hydroxyurea therapy for neurological and cognitive protection in pediatric sickle cell anemia in Uganda (BRAIN SAFE II): Protocol for a single-arm open label trial
- PMID: 39717517
- PMCID: PMC11664125
- DOI: 10.1016/j.conctc.2024.101404
Hydroxyurea therapy for neurological and cognitive protection in pediatric sickle cell anemia in Uganda (BRAIN SAFE II): Protocol for a single-arm open label trial
Abstract
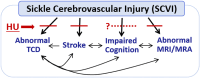
Background: Children with sickle cell anemia (SCA) in Sub-Saharan Africa are at high risk of sickle cerebrovascular injury (SCVI). Hydroxyurea, a commonly used disease-modifying therapy, may reduce SCVI resulting in potential impact on reducing stroke and cognitive dysfunction. We aim to test the impact of daily hydroxyurea therapy on these outcomes in Ugandan children with SCA. We hypothesized that hydroxyurea therapy over 36 months will prevent, stabilize or improve these complications of SCA.
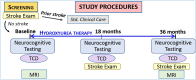
Methods: The BRAIN SAFE II study is an open label, single arm trial of daily hydroxyurea in 270 children with SCA (HbSS) in Uganda, ages 3-9 years. Following baseline assessments, participants began hydroxyurea therapy and are followed according to local guidelines. Standard hydroxyurea dose is escalated to maximum tolerated dose (MTD). SCVI is assessed by cerebral arterial velocity using Doppler ultrasound, with cognitive function determined by formal neurocognitive testing (primary outcomes). Structural SCVI is assessed by magnetic resonance imaging (MRI) and angiography (MRA) in a sub-sample of 90 participants ages >5 years. At trial midpoint (18 months) and completion (36 months), outcomes of age-specific assessments will be compared to baseline, as well as biomarkers of anemia, inflammation and malnutrition (secondary outcomes) to determine their relationships to primary outcomes.
Conclusion: This trial will examine the impact of hydroxyurea on preventing or ameliorating SCA SCVI in children, assessed by reducing incident stroke, stroke risk and neurocognitive dysfunction. Trial results will provide critical insight into the role of hydroxyurea therapy on critical manifestations of SCVI in children with SCA.
Trial registration: https://clinicaltrials.gov/ct2/show/NCT04750707 (registered 11 February 2021).
Protocol version: BRAIN SAFE II Protocol Version 3.0, Mar 02, 2022.
Keywords: Hydroxyurea; Neurological and cognitive protection; Sickle cell anemia; Transcranial Doppler.
© 2024 Published by Elsevier Inc.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Update of
-
Hydroxyurea Therapy for Neurological and Cognitive Protection in Pediatric Sickle Cell Anemia in Uganda (BRAIN SAFE II): Protocol for a single-arm open label trial.medRxiv [Preprint]. 2024 Jan 13:2024.01.12.24301208. doi: 10.1101/2024.01.12.24301208. medRxiv. 2024. Update in: Contemp Clin Trials Commun. 2024 Nov 28;42:101404. doi: 10.1016/j.conctc.2024.101404. PMID: 38260320 Free PMC article. Updated. Preprint.
References
-
- Moeti M., Mocumbi A., Bukhman G. British Medical Journal Publishing Group; 2023. Why There Is New Hope for the Care of Chronic Diseases in Africa. - PubMed
-
- Ndeezi G., Kiyaga C., Hernandez A.G., Munube D., Howard T.A., Ssewanyana I., et al. Burden of sickle cell trait and disease in the Uganda Sickle Surveillance Study (US3): a cross-sectional study. Lancet Global Health. 2016;4(3):e195–e200. - PubMed
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

