Improving PD-1 blockade plus chemotherapy for complete remission of lung cancer by nanoPDLIM2
- PMID: 39718207
- PMCID: PMC11668523
- DOI: 10.7554/eLife.89638
Improving PD-1 blockade plus chemotherapy for complete remission of lung cancer by nanoPDLIM2
Abstract
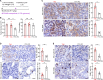
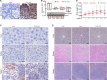
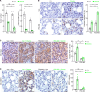
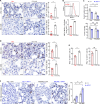
Immune checkpoint inhibitors (ICIs) and their combination with other therapies such as chemotherapy, fail in most cancer patients. We previously identified the PDZ-LIM domain-containing protein 2 (PDLIM2) as a bona fide tumor suppressor that is repressed in lung cancer to drive cancer and its chemo and immunotherapy resistance, suggesting a new target for lung cancer therapy improvement. In this study, human clinical samples and data were used to investigate PDLIM2 genetic and epigenetic changes in lung cancer. Using an endogenous mouse lung cancer model faithfully recapitulating refractory human lung cancer and a clinically feasible nano-delivery system, we investigated the therapeutic efficacy, action mechanism, and safety of systemically administrated PDLIM2 expression plasmids encapsulated in nanoparticles (nanoPDLIM2) and its combination with PD-1 antibody and chemotherapeutic drugs. Our analysis indicate that PDLIM2 repression in human lung cancer involves both genetic deletion and epigenetic alteration. NanoPDLIM2 showed low toxicity, high tumor specificity, antitumor activity, and greatly improved the efficacy of anti-PD-1 and chemotherapeutic drugs, with complete tumor remission in most mice and substantial tumor reduction in the remaining mice by their triple combination. Mechanistically, nanoPDLIM2 increased major histocompatibility complex class I (MHC-I) expression, suppressed multi-drug resistance 1 (MDR1) induction and survival genes and other tumor-related genes expression in tumor cells, and enhanced lymphocyte tumor infiltration, turning the cold tumors hot and sensitive to ICIs and rendering them vulnerable to chemotherapeutic drugs and activated tumor-infiltrating lymphocytes (TILs) including those unleashed by ICIs. These studies established a clinically applicable PDLIM2-based combination therapy with great efficacy for lung cancer and possibly other cold cancers.
Keywords: PD-1; PDLIM2; cancer biology; combination therapy; human; immune checkpoint; lung cancer; mouse; nanotherapy.
Plain language summary
Lung cancer remains the leading cause of all cancer-related deaths. Treatment options are limited because drug-based therapies including chemotherapy and immune checkpoint inhibitors (or ICIs, for short) are ineffective in most patients. PDLIM2 is a protein that normally prevents tumors from forming by regulating the activities of other genes. However, lung cancer cells generally have lower levels of this protein than healthy cells and this appears to be linked to the ability of the cancer cells to become resistant to chemotherapy and ICIs. Cells make proteins using templates encoded in our DNA. It remains unclear how PDLIM2 production is repressed in lung cancer: it is possible that cancer cells may acquire genetic alterations that affect PDLIM2 production, or there may be other changes to the structure of the DNA known as epigenetic changes. Sun et al. investigated the production of PDLIM2 in samples from human lung cancer patients. The experiments found that in over 90% of the patients, the levels of PDLIM2 were lower than in cells from healthy individuals. This was due to genetic alterations or epigenetic changes, or a combination of the two. Further experiments in a mouse model of lung cancer demonstrated that it is possible to use nanotechnology to deliver PDLIM2 to cancer cells for effective cancer therapy with low toxicity. Combining this nanotechnology (known as nanoPDLIM2) with both ICIs and chemotherapy drugs was able to completely eradicate all tumors in most of the mice. The findings provide a firm basis for further studies of the potential of nanoPDLIM2 as a safe and effective therapy for human lung cancer. PDLIM2 production is also repressed in numerous other types of cancer, so it is possible that nanoPDLIM2 may have broader uses in cancer treatment.
© 2023, Sun et al.
Conflict of interest statement
FS, PY, YX, HZ, SS, GX, ZQ No competing interests declared
Figures




Update of
-
Improving PD-1 blockade plus chemotherapy for complete remission of lung cancer by nanoPDLIM2.bioRxiv [Preprint]. 2024 Jan 9:2023.07.23.550248. doi: 10.1101/2023.07.23.550248. bioRxiv. 2024. Update in: Elife. 2024 Dec 24;12:RP89638. doi: 10.7554/eLife.89638. PMID: 37546791 Free PMC article. Updated. Preprint.
References
-
- Buscail L, Bournet B, Vernejoul F, Cambois G, Lulka H, Hanoun N, Dufresne M, Meulle A, Vignolle-Vidoni A, Ligat L, Saint-Laurent N, Pont F, Dejean S, Gayral M, Martins F, Torrisani J, Barbey O, Gross F, Guimbaud R, Otal P, Lopez F, Tiraby G, Cordelier P. First-in-man phase 1 clinical trial of gene therapy for advanced pancreatic cancer: safety, biodistribution, and preliminary clinical findings. Molecular Therapy. 2015;23:779–789. doi: 10.1038/mt.2015.1. - DOI - PMC - PubMed
-
- Commisso C, Davidson SM, Soydaner-Azeloglu RG, Parker SJ, Kamphorst JJ, Hackett S, Grabocka E, Nofal M, Drebin JA, Thompson CB, Rabinowitz JD, Metallo CM, Vander Heiden MG, Bar-Sagi D. Macropinocytosis of protein is an amino acid supply route in Ras-transformed cells. Nature. 2013;497:633–637. doi: 10.1038/nature12138. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

