Exchange, promiscuity, and orthogonality in de novo designed coiled-coil peptide assemblies
- PMID: 39720134
- PMCID: PMC11664599
- DOI: 10.1039/d4sc06329e
Exchange, promiscuity, and orthogonality in de novo designed coiled-coil peptide assemblies
Abstract
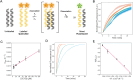
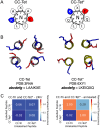
De novo protein design is delivering new peptide and protein structures at a rapid pace. Many of these synthetic polypeptides form well-defined and hyperthermal-stable structures. Generally, however, less is known about the dynamic properties of the de novo designed structures. Here, we explore one aspect of dynamics in a series of de novo coiled-coil peptide assemblies: namely, peptide exchange within and between different oligomers from dimers through to heptamers. First, we develop a fluorescence-based reporter assay for peptide exchange that is straightforward to implement, and, thus, would be useful to others examining similar systems. We apply this assay to explore both homotypic exchange within single species, and heterotypic exchange between coiled coils of different oligomeric states. For the former, we provide a detailed study for a dimeric coiled coil, CC-Di, finding a half-life for exchange of 4.2 ± 0.3 minutes at a peptide concentration of 200 μM. Interestingly, more broadly when assessing exchange across all of the oligomeric states, we find that some of the designs are faithful and only undergo homotypic strand exchange, whereas others are promiscuous and exchange to form unexpected hetero-oligomers. Finally, we develop two design strategies to improve the orthogonality of the different oligomers: (i) using alternate positioning of salt bridge interactions; and (ii) incorporating non-canonical repeats into the designed sequences. In so doing, we reconcile the promiscuity and deliver a set of faithful homo-oligomeric de novo coiled-coil peptides. Our findings have implications for the application of these and other coiled coils as modules in chemical and synthetic biology.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures




References
LinkOut - more resources
Full Text Sources

