The role of amphipathic and cationic helical peptides in Parkinson's disease
- PMID: 39720890
- PMCID: PMC11669119
- DOI: 10.1002/pro.70020
The role of amphipathic and cationic helical peptides in Parkinson's disease
Abstract
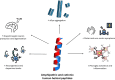
Peptides are attracting a growing interest for therapeutic applications in biomedicine. In Parkinson's disease (PD), different human endogenous peptides have been associated with beneficial effects, including protein aggregation inhibition, reduced inflammation, or the protection of dopaminergic neurons. Such effects seem to be connected to the spatial arrangement of peptide side chains, and many of these human molecules share common conformational traits, displaying a distinctive amphipathic and cationic helical structure, which is believed to be crucial for their activities. This review delves into the relationship between these structural properties and the current evidence connecting biogenic peptides to the amelioration of PD symptoms. We discuss their implications in the disease, the different mechanisms of action, their state of validation, and their therapeutic potential.
Keywords: Parkinson's disease; peptides; structure; therapy; α‐synuclein.
© 2024 The Author(s). Protein Science published by Wiley Periodicals LLC on behalf of The Protein Society.
Figures

References
-
- Abuirmeileh A, Lever R, Kingsbury AE, Lees AJ, Locke IC, Knight RA, et al. The corticotrophin‐releasing factor‐like peptide urocortin reverses key deficits in two rodent models of Parkinson's disease. Eur J Neurosci. 2007;26(2):417–423. - PubMed
-
- Ago Y, Hayata A, Hashimoto H. Pathophysiological implication of the VPAC2 receptor in psychiatric disorders. Nihon Yakurigaku Zasshi. 2018;151(6):249–253. - PubMed
-
- Arimura A. Perspectives on pituitary adenylate cyclase activating polypeptide (PACAP) in the neuroendocrine, endocrine, and nervous systems. Jpn J Physiol. 1998;48(5):301–331. - PubMed
-
- Arimura A, Somogyvari‐Vigh A, Weill C, Fiore RC, Tatsuno I, Bay V, et al. Pacap functions as a neurotrophic factor. Ann N Y Acad Sci. 1994;739:228–243. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

