Membrane nanodomains to shape plant cellular functions and signaling
- PMID: 39722237
- PMCID: PMC11754938
- DOI: 10.1111/nph.20367
Membrane nanodomains to shape plant cellular functions and signaling
Abstract
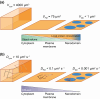
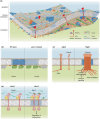
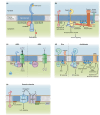
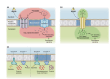
Plasma membrane (PM) nanodomains have emerged as pivotal elements in the regulation of plant cellular functions and signal transduction. These nanoscale membrane regions, enriched in specific lipids and proteins, behave as regulatory/signaling hubs spatially and temporally coordinating critical cellular functions. In this review, we first examine the mechanisms underlying the formation and maintenance of PM nanodomains in plant cells, highlighting the roles of PM lipid composition, protein oligomerization and interactions with cytoskeletal and cell wall components. Then, we discuss how nanodomains act as organizing centers by mediating protein-protein interactions that orchestrate essential processes such as symbiosis, defense against pathogens, ion transport or hormonal and reactive oxygen species (ROS) signaling. Finally, we introduce the concept of nanoenvironments, where localized physicochemical variations are generated in the very close proximity of PM nanodomains, in response to stimuli. After decoding by a dedicated machinery likely localized in the vicinity of nanodomains, this enrichment of secondary messengers, such as ROS or Ca2+, would allow specific downstream cellular responses. This review provides insights into the dynamic nature of nanodomains and proposes future research to better understand their contribution to the intricate signaling networks that govern plant development and stress responses.
Keywords: lipids; nanodomain; nanoenvironment; plant; plasma membrane; protein–protein interactions; signaling.
© 2024 The Author(s). New Phytologist © 2024 New Phytologist Foundation.
Conflict of interest statement
None declared.
Figures
References
-
- Albers P, Üstün S, Witzel K, Kraner M, Börnke F. 2019. A Remorin from Nicotiana benthamiana interacts with the Pseudomonas type‐III effector protein HopZ1a and is phosphorylated by the immune‐related kinase PBS1. Molecular Plant–Microbe Interactions 32: 1229–1242. - PubMed
-
- Aryal B, Xia J, Hu Z, Stumpe M, Tsering T, Liu J, Huynh J, Fukao Y, Glöckner N, Huang H‐Y et al. 2023. An LRR receptor kinase controls ABC transporter substrate preferences during plant growth‐defense decisions. Current Biology 33: 2008–2023. - PubMed
-
- Attacha S, Solbach D, Bela K, Moseler A, Wagner S, Schwarzländer M, Aller I, Müller SJ, Meyer AJ. 2017. Glutathione peroxidase‐like enzymes cover five distinct cell compartments and membrane surfaces in Arabidopsis thaliana . Plant, Cell & Environment 40: 1281–1295. - PubMed
-
- Augustine GJ, Adler EM, Charlton MP, Hans M, Swandulla D, Zipser K. 1992. Presynaptic calcium signals during neurotransmitter release: detection with fluorescent indicators and other calcium chelators. Journal of Physiology 86: 129–134. - PubMed
-
- Bahammou D, Recorbet G, Mamode Cassim A, Robert F, Balliau T, Van Delft P, Haddad Y, Mongrand S, Fouillen L, Simon‐Plas F. 2024. A combined lipidomic and proteomic profiling of Arabidopsis thaliana plasma membrane. The Plant Journal 119: 1570–1595. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

