Clinical Insights Into Default Mode Network Abnormalities in Mild Traumatic Brain Injury: Unraveling Axonal Injury Through Functional, Structural, and Molecular Analyses
- PMID: 39722654
- PMCID: PMC11669841
- DOI: 10.1111/cns.70188
Clinical Insights Into Default Mode Network Abnormalities in Mild Traumatic Brain Injury: Unraveling Axonal Injury Through Functional, Structural, and Molecular Analyses
Abstract
Background: Mild traumatic brain injury (mTBI) frequently results in persistent cognitive, emotional, and functional impairments, closely linked to disruptions in the default mode network (DMN). Understanding the mechanisms driving these network abnormalities is critical for advancing diagnostic and therapeutic strategies.
Methods: This study adopted a multimodal approach, combining functional connectivity (FC) analysis, diffusion tensor imaging (DTI), and gene expression profiling to investigate DMN disruptions in mTBI. A primary focus was placed on the middle cingulate cortex (MCC), a region consistently identified with increased connectivity. We explored the structural and molecular changes underlying this phenomenon. Receiver operating characteristic (ROC) curve analysis was utilized to assess the diagnostic potential of DTI-derived metrics, while white matter tractography was employed to explore structural connectivity between the MCC and the dorsolateral prefrontal cortex (DLPFC).
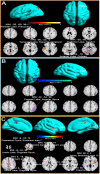
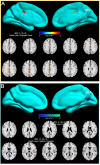
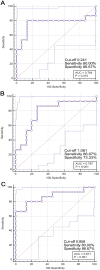
Results: Our findings revealed significant disruptions in DMN connectivity, with the MCC prominently involved in mTBI pathology. DTI analyses identified pronounced axonal injury in the MCC, characterized by decreased fractional anisotropy (FA) and axial diffusivity (AD), alongside increased isotropy (ISO), indicating compromised white matter integrity and diffuse axonal injury. Gene expression profiling revealed the upregulation of pathways related to synaptic transmission, ion channel regulation, and axonal injury response. ROC analysis demonstrated that ISO serves as a particularly effective biomarker for mTBI, showing high diagnostic accuracy (AUC = 0.871). White matter tractography further confirmed strong structural connectivity between the MCC and the DLPFC, identifying potential therapeutic targets for neuromodulation.
Conclusion: This study provides robust evidence that diffuse axonal injury plays a pivotal role in DMN abnormalities observed in mTBI. The integration of FC, DTI, and gene expression profiling offers a comprehensive framework for understanding mTBI's impact on brain networks. Our findings also highlight the DLPFC as a promising target for therapeutic interventions aimed at addressing cognitive and emotional deficits associated with mTBI.
Keywords: axonal injury; default mode network; diffusion tensor imaging; functional connectivity; mild traumatic brain injury.
© 2024 The Author(s). CNS Neuroscience & Therapeutics published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



References
-
- Visser K., Koggel M., Blaauw J., van der Horn H. J., Jacobs B., and van der Naalt J., “Blood‐Based Biomarkers of Inflammation in Mild Traumatic Brain Injury: A Systematic Review,” Neuroscience and Biobehavioral Reviews 132 (2022): 154–168. - PubMed
Publication types
MeSH terms
Grants and funding
- 2022-WS-05/The Shanghai Jinshan District Science and Technology Innovation Fund Project
- JSZK2023B01/The Shanghai Jinshan District Health System Medical Key Specialty Construction Project
- 82171382/The National Natural Science Foundation of China
- 82471407/The National Natural Science Foundation of China
- SHDC22023224/The Shanghai Shenkang Hospital Development Center Municipal Hospital Diagnosis and Treatment Technology Promotion and Optimization Management Project
LinkOut - more resources
Full Text Sources
Medical

