Developmental deglutition and intrinsic tongue muscle maturation phenotypes in the Ts65Dn mouse model of Down syndrome
- PMID: 39722691
- PMCID: PMC11668655
- DOI: 10.3389/fneur.2024.1461682
Developmental deglutition and intrinsic tongue muscle maturation phenotypes in the Ts65Dn mouse model of Down syndrome
Abstract
Introduction: Down syndrome (DS) is associated with difficulties with feeding during infancy and childhood. Weaning, or transitioning from nursing to independent deglutition, requires developmental progression in tongue function. However, little is known about whether postnatal tongue muscle maturation is impacted in DS. This study tested the hypothesis that the Ts65Dn mouse model of DS has developmental delays in deglutition, comprised of differences in eating and drinking behaviors relative to euploid controls, coinciding with atypical measures of intrinsic tongue muscle microanatomy.
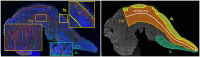
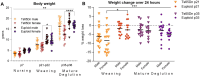
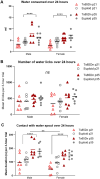
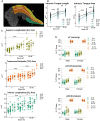
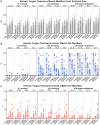
Methods: The Ts65Dn mouse model of DS and euploid controls were evaluated at 7 days of age (p7; nursing), p21 (weaning), and p35 (mature deglutition) (n = 13-18 mice per group). Eating behavior, drinking behavior, and body weight changes were quantified in p21 and p35 mice through the use of automated monitoring over 24 h. Intrinsic tongues of mice at all three ages were sectioned and stained to permit quantification of the sizes of the four major intrinsic tongue muscles. Transverse intrinsic tongue muscles were evaluated for myofiber size (average myofiber cross sectional area (CSA) of all fibers, MyHC2a fibers, MyHC 2b fibers, and minimum Feret fiber diameter), and percentage of MyHC isoforms (%MyHC2a + fibers, and %MyHC 2b + fibers) in anterior, middle, and posterior regions.
Results: Ts65Dn showed significant differences from euploid in deglutition measures. Compared to euploid, Ts65Dn also showed differences in intrinsic tongue muscle microanatomy and biology. Specifically, Ts65Dn intrinsic tongues had smaller transverse muscle myofiber size measures than control in the anterior and middle tongue, but not in the posterior tongue.
Conclusion: Differences in intrinsic tongue muscles coincide with feeding phenotypes in the Ts65Dn mouse model of DS.
Keywords: Down syndrome; Ts65Dn; deglutition; feeding; intrinsic tongue; maturation; mouse; weaning.
Copyright © 2024 Glass, Chatwin, Fisher, Hang, Yang, Brutto, Waghray and Connor.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Physical exercise training interventions for children and young adults during and after treatment for childhood cancer.Cochrane Database Syst Rev. 2016 Mar 31;3(3):CD008796. doi: 10.1002/14651858.CD008796.pub3. Cochrane Database Syst Rev. 2016. PMID: 27030386 Free PMC article.
-
Sertindole for schizophrenia.Cochrane Database Syst Rev. 2005 Jul 20;2005(3):CD001715. doi: 10.1002/14651858.CD001715.pub2. Cochrane Database Syst Rev. 2005. PMID: 16034864 Free PMC article.
-
Home treatment for mental health problems: a systematic review.Health Technol Assess. 2001;5(15):1-139. doi: 10.3310/hta5150. Health Technol Assess. 2001. PMID: 11532236
-
Corticosteroids for the treatment of Duchenne muscular dystrophy.Cochrane Database Syst Rev. 2016 May 5;2016(5):CD003725. doi: 10.1002/14651858.CD003725.pub4. Cochrane Database Syst Rev. 2016. PMID: 27149418 Free PMC article.
-
Sexual Harassment and Prevention Training.2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 36508513 Free Books & Documents.
References
-
- Donnelly LF, Shott SR, LaRose CR, Chini BA, Amin RS. Causes of persistent obstructive sleep apnea despite previous tonsillectomy and adenoidectomy in children with Down syndrome as depicted on static and dynamic cine MRI. AJR Am J Roentgenol. (2004) 183:175–81. doi: 10.2214/ajr.183.1.1830175, PMID: - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

