An exploratory study of the safety profile and neurocognitive function after single doses of mitragynine in humans
- PMID: 39724441
- PMCID: PMC12084173
- DOI: 10.1007/s00213-024-06734-2
An exploratory study of the safety profile and neurocognitive function after single doses of mitragynine in humans
Abstract
Rationale: Despite the growing scientific interest on mitragynine, the primary alkaloid in kratom (Mitragyna Speciosa), there is a lack of clinical trials in humans.
Objectives: This phase 1 study aimed to evaluate mitragynine's safety profile and acute effects on subjective drug experience, neurocognition, and pain tolerance.
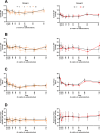
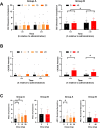
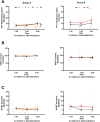
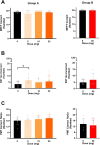
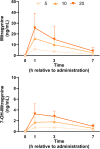
Methods: A placebo-controlled, single-blind, within-subjects study was conducted in two parts. In part A, eight healthy human volunteers received placebo and three doses of mitragynine (5, 10, and 20 mg) in a sequential dosing scheme, on separate days. In part B, a second group of seven volunteers received placebo and 40 mg of mitragynine. Vital signs, subjective drug experience, neurocognitive function, and pain tolerance were measured at regular intervals for 7 h after administration.
Results: Overall, mitragynine did not affect most of the outcome measures at any dose. Yet, the lowest dose (5 mg) of mitragynine increased subjective ratings of arousal and attention, accuracy in a sustained attention task, and motor inhibition. The highest dose (40 mg) of mitragynine increased subjective ratings of amnesia and produced mild psychopathological symptoms. Mitragynine did not significantly affect vital signs, and only mild, transient side effects were reported.
Conclusion: The present study suggests that low doses (5-10 mg) of mitragynine may cause subjective feelings of stimulation and enhance attention, while the highest dose (40 mg) may cause inhibitory feelings of amnesia and distress. Mitragynine doses up to 40 mg were well tolerated in this group.
Keywords: Mitragyna speciosa; Acute effects; Kratom; Mitragynine; Neurocognition; Pain; Placebo-controlled study; Safety.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Conflict of interest: None.
Figures
References
-
- Alsarraf E, Myers J, Culbreth S, Fanikos J (2019) Kratom from head to toe—case reviews of adverse events and toxicities. Curr Emerg Hosp Med Rep 7:141–168 - DOI
-
- Barratt ES (1993) Impulsivity: Integrating cognitive, behavioral, biological, and environmental data
-
- Bremner J (2014) The clinician administered dissociative states scale (CADSS): instructions for administration. Emory University
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

