SARS-CoV-2 SPIKE Antibody Levels can Indicate Immuno-Resilience to Re-infection: a Real-World Study
- PMID: 39724513
- PMCID: PMC11782789
- DOI: 10.1007/s40121-024-01090-2
SARS-CoV-2 SPIKE Antibody Levels can Indicate Immuno-Resilience to Re-infection: a Real-World Study
Abstract
Introduction: The use of antibody titers against SARS-CoV-2, as a method of estimating subsequent infection following infection or vaccination, is unclear. Here, we investigate whether specific levels of antibodies, as markers of adaptive immunity, can serve to estimate the risk of symptomatic SARS-CoV-2 (re-) infection.
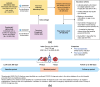
Methods: In this real-world study, laboratory data from individuals tested for SARS-CoV-2 antibodies under routine clinical conditions were linked through tokenization to a United States medical insurance claims database to determine the risk of symptomatic/severe SARS-CoV-2 infection outcomes. Antibody titer levels were determined using the Elecsys® Anti-SARS-CoV-2 S assay. Study outcomes included the first symptomatic SARS-CoV-2 infection (per ICD-10 diagnostic codes, occurring ≥ 7 days post-antibody titer test), and severe SARS-CoV-2 infection, characterized by adverse outcomes including hospitalization, intensive care unit admission, intubation, mechanical ventilation, or death within 30 days of infection. All outcomes were assessed for 12 months following antibody measurement. Hazard ratios of subsequent symptomatic and severe infections were estimated using Cox regression with inverse probability weighting.
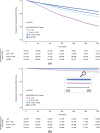
Results: Of 268,844 individuals with antibody data (April 2021-June 2022), those with levels ≥ 0.8 to < 1,000 U/mL had a 42% reduced risk of symptomatic infection within 12 months, compared with < 0.8 U/mL (HR = 0.58, 95% CI [0.55, 0.61]). The risk decreased by 53% (HR = 0.47, 95% CI [0.45, 0.49]) with ≥ 1000 to < 2500 U/mL and by 62% (HR = 0.38 [0.36, 0.39]) for ≥ 2500 U/mL. Risk of severe SARS-CoV-2 outcomes was also reduced. Subgroup analyses showed a consistent association between antibody levels and infection risk, by immune status and age. Clinically meaningful thresholds of antibody titers varied between Delta and Omicron infections.
Conclusion: Higher antibody titer levels indicated reduced risk of developing symptomatic or severe COVID-19. Titers of ≥ 2500 U/mL indicated a 62-87% reduced infection risk. The quantitative determination of antibody titers allowed scaling of the correlate of risk to new variants.
Keywords: COVID-19; Immune response; Immunity; Real-world data; Risk estimate; SARS-CoV-2; Spike antibody titer.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Conflict of interest: Dr Yue Jin is an employee of Roche Molecular Systems Inc. and a Roche stockholder; Dr Fei Yang is currently an employee of Phillip Morris International but was an employee of Roche Information Solutions Ltd at the time the study was conducted. Phillip Morris International has not been involved in the study or publication; Dr Christopher M. Rank is an employee of Roche Diagnostics GmbH and holds stocks/shares in F. Hofmann-La Roche Ltd; Dr Stanley Letovsky is a Labcorp employee and participates in employee stock plans; Dr Peter Ramge is an employee of Roche Diagnostics International Ltd. and a Roche stockholder; Dr Simon Jochum is an employee of Roche Diagnostics GmbH. The Elecsys Anti-SARS-CoV-2 S assay is approved under an Emergency Use Authorization in the US. ELECSYS is a trademark of Roche. All other product names and trademarks are the property of their respective owners. Ethical Approval: As a non-interventional study based on secondary data use, this study did not require informed consent or institutional/ethical review board approval.
Figures

Similar articles
-
Wild-type SARS-CoV-2 neutralizing immunity decreases across variants and over time but correlates well with diagnostic testing.Front Immunol. 2023 Feb 8;14:1055429. doi: 10.3389/fimmu.2023.1055429. eCollection 2023. Front Immunol. 2023. PMID: 36845123 Free PMC article.
-
Bimodal antibody-titer decline following BNT162b2 mRNA anti-SARS-CoV-2 vaccination in healthcare workers of the INT - IRCCS "Fondazione Pascale" Cancer Center (Naples, Italy).Infect Agent Cancer. 2022 Jul 28;17(1):40. doi: 10.1186/s13027-022-00451-1. Infect Agent Cancer. 2022. PMID: 35902961 Free PMC article.
-
Antibody titer levels and the effect on subsequent SARS-CoV-2 infection in a large US-based cohort.Heliyon. 2023 Feb;9(2):e13103. doi: 10.1016/j.heliyon.2023.e13103. Epub 2023 Jan 21. Heliyon. 2023. PMID: 36712916 Free PMC article.
-
SARS-CoV-2 spike-protein targeted serology test results and their association with subsequent COVID-19-related outcomes.Front Public Health. 2023 Jul 25;11:1193246. doi: 10.3389/fpubh.2023.1193246. eCollection 2023. Front Public Health. 2023. PMID: 37559735 Free PMC article.
-
Anti-SARS-CoV-2 serology based on ancestral RBD antigens does not correlate with the presence of neutralizing antibodies against Omicron variants.Microbiol Spectr. 2025 Jan 7;13(1):e0156824. doi: 10.1128/spectrum.01568-24. Epub 2024 Nov 20. Microbiol Spectr. 2025. PMID: 39565133 Free PMC article.
References
-
- Khoury DS, Cromer D, Reynaldi A, Schlub TE, Wheatley AK, Juno JA, et al. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat Med. 2021;27(7):1205–11. - PubMed
-
- Kaufman HW, Meyer WA, Clarke NJ, Radcliff J, Rank CM, Freeman J, et al. Assessing vulnerability to COVID-19 in high-risk populations: the role of SARS-CoV-2 Spike-targeted serology. Popul Health Manag. 2023;26(1):29–36. - PubMed
-
- Mink S, List W, Hoefle G, Frick M, Suessenbacher A, Winder T, et al. Evaluation of SARS-CoV-2 antibody levels on hospital admission as a correlate of protection against mortality. J Intern Med. 2023;293(6):694–703. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

