Design, Synthesis, and Evaluation of Novel (-)-cis-N-Normetazocine Derivatives: In Vitro and Molecular Modeling Insights
- PMID: 39726303
- PMCID: PMC11671793
- DOI: 10.1111/cbdd.70037
Design, Synthesis, and Evaluation of Novel (-)-cis-N-Normetazocine Derivatives: In Vitro and Molecular Modeling Insights
Abstract
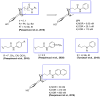
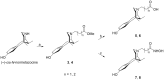
Suitable structural modifications of the functional groups at N-substituent of (-)-cis-N-normetazocine nucleus modulate the affinity and activity profile of related ligands toward opioid receptors. Our research group has developed several compounds and the most interesting ligands, LP1 and LP2, exhibited a dual-target profile for mu-opioid receptor (MOR) and delta-opioid receptor (DOR). Recent structure-affinity relationship studies led to the discovery of novel LP2 analogs (compounds 1 and 2), which demonstrated high MOR affinity in the nanomolar range. Here, we reported the synthesis of the new (-)-cis-N-normetazocine derivatives (3-8) characterized by the absence of the phenyl ring in the N-substituent compared to all previous reported ligands. Compounds 3 and 4, featuring a methyl ester functional group in the N-substituent, retained significant MOR affinity and exhibited similar affinity for the kappa-opioid receptor (KOR). In contrast, compounds 7 and 8, which contain a hydroxamic acid functionality, maintained affinity exclusively toward MOR. Neither of compounds (3-8) showed DOR affinity. Molecular modeling studies confirmed a similar docking pose in the MOR binding pocket for these compounds. Additionally, the in silico ADME profile of the most interesting ligands (3, 4, 7, and 8) was investigated revealing a favorable profile for compound 7 regarding the blood-brain barrier permeability, suggesting its potential as a peripherally restricted opioid ligand.
Keywords: ADME studies; cis‐N‐normetazocine; docking studies; mu opioid receptor; radioligand competition‐binding.
© 2024 The Author(s). Chemical Biology & Drug Design published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


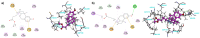
Similar articles
-
Synthesis and Structure-Activity Relationships of (-)-cis-N-Normetazocine-Based LP1 Derivatives.Pharmaceuticals (Basel). 2018 May 5;11(2):40. doi: 10.3390/ph11020040. Pharmaceuticals (Basel). 2018. PMID: 29734749 Free PMC article.
-
Design, synthesis, in vitro evaluation, and molecular modeling studies of N-substituted benzomorphans, analogs of LP2, as novel MOR ligands.Chem Biol Drug Des. 2023 Jun;101(6):1382-1392. doi: 10.1111/cbdd.14220. Epub 2023 Mar 5. Chem Biol Drug Des. 2023. PMID: 36813756
-
Development of novel LP1-based analogues with enhanced delta opioid receptor profile.Bioorg Med Chem. 2017 Sep 1;25(17):4745-4752. doi: 10.1016/j.bmc.2017.07.021. Epub 2017 Jul 11. Bioorg Med Chem. 2017. PMID: 28734666
-
Synthesis and Structure-Activity Relationships of LP1 Derivatives: N-Methyl-N-phenylethylamino Analogues as Novel MOR Agonists.Molecules. 2018 Mar 16;23(3):677. doi: 10.3390/molecules23030677. Molecules. 2018. PMID: 29547588 Free PMC article.
-
LP1 and LP2: Dual-Target MOPr/DOPr Ligands as Drug Candidates for Persistent Pain Relief.Molecules. 2021 Jul 8;26(14):4168. doi: 10.3390/molecules26144168. Molecules. 2021. PMID: 34299443 Free PMC article. Review.
References
-
- Brine, G. A. , Berrang B., Hayes J. P., and Carroll F. I.. 1990. “An Improved Resolution of (+/−)‐cis‐N‐Normetazocine.” Journal of Heterocyclic Chemistry 27, no. 7: 2139–2143. 10.1002/jhet.5570270753. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

