Rituximab Safety Profile: A Seven-Year Retrospective Analysis From Southern India
- PMID: 39726516
- PMCID: PMC11669735
- DOI: 10.7759/cureus.74454
Rituximab Safety Profile: A Seven-Year Retrospective Analysis From Southern India
Abstract
Objective: This study aimed to analyze the pattern, severity, and outcomes of adverse drug reactions (ADRs) associated with rituximab use reported to a regional pharmacovigilance center in Kerala, India.
Methods: This retrospective study analyzed rituximab-associated ADRs reported from 2017 to 2023. ADRs were assessed using the WHO-UMC criteria for causality and the Modified Hartwig Siegel Scale for severity.
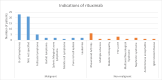
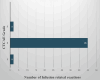
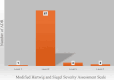
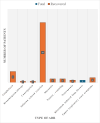
Results: A total of 74 patients reported ADRs, with 49 being female. The majority (27 patients) were in the 51- to 60-year age group. Malignancies accounted for 58 cases of rituximab use. Most ADRs (69 cases) occurred with the first dose, with infusion reactions being the most common. Causality assessment revealed that 48 ADRs were probably related to rituximab. Severity analysis using the Modified Hartwig Siegel Scale showed that 67 ADRs were level two (mild), while two were level seven (severe). The majority of patients (72) recovered. However, rituximab-induced lung disease, observed in two cases, was associated with higher mortality.
Conclusion: Rituximab demonstrated an acceptable safety profile, with most ADRs being non-serious and manageable. The high recovery rate reflects effective ADR management. However, the potential for serious complications, particularly rituximab-induced lung disease, highlights the need for vigilant monitoring, especially during the first infusions. These findings enhance understanding of rituximab's real-world safety profile and underscore the importance of standardized protocols for ADR mitigation.
Keywords: adverse drug reactions; causality assessment; infusion related reactions; modified hartwig siegel scale; pharmacovigilance; rituximab; rituximab-induced lung disease; safety profile; who-umc criteria.
Copyright © 2024, Jayasree et al.
Conflict of interest statement
Human subjects: Consent for treatment and open access publication was obtained or waived by all participants in this study. Ethics Committee of Amrita School of Medicine issued approval ECASM-AIMS-2024-403. Animal subjects: All authors have confirmed that this study did not involve animal subjects or tissue. Conflicts of interest: In compliance with the ICMJE uniform disclosure form, all authors declare the following: Payment/services info: All authors have declared that no financial support was received from any organization for the submitted work. Financial relationships: All authors have declared that they have no financial relationships at present or within the previous three years with any organizations that might have an interest in the submitted work. Other relationships: All authors have declared that there are no other relationships or activities that could appear to have influenced the submitted work.
Figures
Similar articles
-
Pharmacovigilance Study of Anticancer Drugs in a Tertiary Care Teaching Hospital in North India: A Retrospective Study.Cureus. 2023 Sep 10;15(9):e44984. doi: 10.7759/cureus.44984. eCollection 2023 Sep. Cureus. 2023. PMID: 37822427 Free PMC article.
-
Pharmacovigilance in Geriatrics: Exploration of Etio-Clinical Profile and Severity of Adverse Drug Reactions at a Tertiary Hospital in South India.Curr Drug Saf. 2025 Mar 25. doi: 10.2174/0115748863337565250312075339. Online ahead of print. Curr Drug Saf. 2025. PMID: 40143695
-
Study of pattern & distribution of adverse drug reactions in acute coronary syndrome patients in a tertiary care hospital.Indian J Med Res. 2022 Jul;156(1):111-121. doi: 10.4103/ijmr.IJMR_1275_20. Indian J Med Res. 2022. PMID: 36510903 Free PMC article.
-
Causality, Severity, Preventability and Predictability Assessments Scales for Adverse Drug Reactions: A Review.Cureus. 2024 May 9;16(5):e59975. doi: 10.7759/cureus.59975. eCollection 2024 May. Cureus. 2024. PMID: 38854273 Free PMC article. Review.
-
Current Scenario and Future Prospects of Adverse Drug Reactions (ADRs) Monitoring and Reporting Mechanisms in the Rural Areas of India.Curr Drug Saf. 2024;19(2):172-190. doi: 10.2174/1574886318666230428144120. Curr Drug Saf. 2024. PMID: 37132145 Review.
References
-
- Past, present, and future of rituximab-the world’s first oncology monoclonal antibody therapy. Pierpont TM, Limper CB, Richards KL. https://www.frontiersin.org/journals/oncology/articles/10.3389/fonc.2018.... Front Oncol. 2018;8:163. - PMC - PubMed
-
- Addition of rituximab to fludarabine and cyclophosphamide in patients with chronic lymphocytic leukaemia: a randomised, open-label, phase 3 trial. Hallek M, Fischer K, Fingerle-Rowson G, et al. Lancet. 2010;376:1164–1174. - PubMed
-
- CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. Coiffier B, Lepage E, Briere J, et al. https://www.nejm.org/doi/full/10.1056/NEJMoa011795. N Engl J Med. 2002;346:235–242. - PubMed
-
- Depletion of B cells in vivo by a chimeric mouse human monoclonal antibody to CD20. Reff ME, Carner K, Chambers KS, et al. https://www.sciencedirect.com/science/article/pii/S000649712077669X?via%.... Blood. 1994;83:435–445. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
