Acceptance and safety of the RSV-preventive treatment of newborns with nirsevimab in the maternity department: a prospective longitudinal cohort study in France
- PMID: 39726670
- PMCID: PMC11669793
- DOI: 10.1016/j.eclinm.2024.102986
Acceptance and safety of the RSV-preventive treatment of newborns with nirsevimab in the maternity department: a prospective longitudinal cohort study in France
Abstract
Background: To evaluate the acceptance and safety of the treatment of newborns with nirsevimab (a long-acting monoclonal antibody designed to prevent respiratory syncytial virus infections) during the first season of implementation.
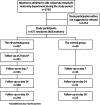
Methods: A longitudinal, prospective, single-centre cohort study was conducted from September 18th, 2023, to January 23rd, 2024 at Lille University Hospital (Lille, France). All newborns admitted to the hospital's maternity department during the study period and whose parents agreed to participate in the study were included. Parents were asked to state whether or not they agreed for their infant to receive nirsevimab. The occurrence of adverse events (AEs) 2 h after nirsevimab treatment and 7, 14 and 30 days after discharge was documented by the mother. The primary endpoint was the nirsevimab treatment acceptance rate. The secondary endpoints were the variables associated with the acceptance of nirsevimab, the reasons for accepting or refusing nirsevimab, and the treatment's real-life safety, relative to a non-treated control group of newborns.
Findings: Of the 1730 infants born in the hospital during the study period, 477 met all the inclusion criteria and were enrolled. The nirsevimab acceptance rate [95% confidence interval] was 91.6% [89.1%-94.2%]. In a multivariable analysis, the mother's age, lower parity and having a partner in work were significantly associated with nirsevimab acceptance. The most common reason for accepting treatment was "to protect my baby", and the most common reason for refusing treatment was the lack of long-term data on nirsevimab. The nirsevimab and control groups did not differ significantly in terms of the types and frequencies of AEs. At least one serious AE was reported for 9.4% of the infants in the nirsevimab group and for 10.3% in the control group. None of the serious AEs were considered to be related to nirsevimab treatment.
Interpretation: The nirsevimab acceptance rate for newborns in the maternity unit was high during the first season of implementation. The safety profile was very good, with no significant differences between the nirsevimab group and the control group.
Funding: None.
Keywords: Acceptance; Newborn; Nirsevimab; Respiratory syncytial virus; Safety.
© 2024 The Author(s).
Conflict of interest statement
FD was member of a board expert about VRS for Sanofi-Pasteur and about vaccines in children for MSD. FD has been invited at the ESPID meeting by MSD in 2024 to present preliminary results of this research. MLa was member of a board expert about meningococcus for Sanofi-Pasteur. Other authors have no conflict of interest to declare in relation to this research.
References
-
- Du Y., Yan R., Wu X., et al. Global burden and trends of respiratory syncytial virus infection across different age groups from 1990 to 2019: a systematic analysis of the Global Burden of Disease 2019 Study. Int J Infect Dis. 2023;135:70–76. - PubMed
-
- Hall C.B., Weinberg G.A., Blumkin A.K., et al. Respiratory syncytial virus–associated hospitalizations among children less than 24 Months of age. Pediatrics. 2013;132:e341–e348. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous