Impact of storage and extraction methods on peat soil microbiomes
- PMID: 39726749
- PMCID: PMC11670759
- DOI: 10.7717/peerj.18745
Impact of storage and extraction methods on peat soil microbiomes
Abstract
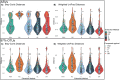
Recovered microbial community structure is known to be influenced by sample storage conditions and nucleic acid extraction methods, and the impact varies by sample type. Peat soils store a large portion of soil carbon and their microbiomes mediate climate feedbacks. Here, we tested three storage conditions and five extraction protocols on peat soils from three physicochemically distinct habitats in Stordalen Mire, Sweden, revealing significant methodological impacts on microbial (here, meaning bacteria and archaea) community structure. Initial preservation method impacted alpha but not beta diversity, with in-field storage in LifeGuard buffer yielding roughly two-thirds the richness of in-field flash-freezing or transport from the field on ice (all samples were stored at -80 °C after return from the field). Nucleic acid extraction method impacted both alpha and beta diversity; one method (the PowerSoil Total RNA Isolation kit with DNA Elution Accessory kit) diverged from the others (PowerMax Soil DNA Isolation kit-High Humic Acid Protocol, and three variations of a modified PowerMax Soil DNA/RNA isolation kit), capturing more diverse microbial taxa, with divergent community structures. Although habitat and sample depth still consistently dominated community variation, method-based biases in microbiome recovery for these climatologically-relevant soils are significant, and underscore the importance of methodological consistency for accurate inter-study comparisons, long-term monitoring, and consistent ecological interpretations.
Keywords: Extraction; Methods; Microbiome; Peatland; Soil; Storage; Stordalen.
© 2024 Cronin et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures




References
-
- Albers CN, Jensen A, Bælum J, Jacobsen CS. Inhibition of DNA polymerases used in Q-PCR by structurally different soil-derived humic substances. Geomicrobiology Journal. 2013;30(8):675–681. doi: 10.1080/01490451.2012.758193. - DOI
-
- Andrews S. FastQC: a quality control tool for high throughput sequence data. 2010. https://www.bioinformatics.babraham.ac.uk/projects/fastqc/ https://www.bioinformatics.babraham.ac.uk/projects/fastqc/
-
- Apprill A, McNally S, Parsons R, Weber L. Minor revision to V4 region SSU rRNA 806R gene primer greatly increases detection of SAR11 bacterioplankton. Aquatic Microbial Ecology. 2015;75(2):129–137. doi: 10.3354/ame01753. - DOI
-
- Bolyen E, Rideout JR, Dillon MR, Bokulich NA, Abnet CC, Al-Ghalith GA, Alexander H, Alm EJ, Arumugam M, Asnicar F, Bai Y, Bisanz JE, Bittinger K, Brejnrod A, Brislawn CJ, Brown CT, Callahan BJ, Caraballo-Rodríguez AM, Chase J, Cope EK, Da Silva R, Diener C, Dorrestein PC, Douglas GM, Durall DM, Duvallet C, Edwardson CF, Ernst M, Estaki M, Fouquier J, Gauglitz JM, Gibbons SM, Gibson DL, Gonzalez A, Gorlick K, Guo J, Hillmann B, Holmes S, Holste H, Huttenhower C, Huttley GA, Janssen S, Jarmusch AK, Jiang L, Kaehler BD, Kang KB, Keefe CR, Keim P, Kelley ST, Knights D, Koester I, Kosciolek T, Kreps J, Langille MGI, Lee J, Ley R, Liu Y-X, Loftfield E, Lozupone C, Maher M, Marotz C, Martin BD, McDonald D, McIver LJ, Melnik AV, Metcalf JL, Morgan SC, Morton JT, Naimey AT, Navas-Molina JA, Nothias LF, Orchanian SB, Pearson T, Peoples SL, Petras D, Preuss ML, Pruesse E, Rasmussen LB, Rivers A, Robeson MS, Rosenthal P, Segata N, Shaffer M, Shiffer A, Sinha R, Song SJ, Spear JR, Swafford AD, Thompson LR, Torres PJ, Trinh P, Tripathi A, Turnbaugh PJ, Ul-Hasan S, Van Der Hooft JJJ, Vargas F, Vázquez-Baeza Y, Vogtmann E, Von Hippel M, Walters W, Wan Y, Wang M, Warren J. Reproducible, interactive, scalable and extensible microbiome data science using QIIME2. Nature Biotechnology. 2019;37(8):852–857. doi: 10.1038/s41587-019-0209-9. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

