How Chromatin Motor Complexes Influence the Nuclear Architecture: A Review of Chromatin Organization, Cohesins, and Condensins with a Focus on C. elegans
- PMID: 39726802
- PMCID: PMC11671135
- DOI: 10.3390/dna4010005
How Chromatin Motor Complexes Influence the Nuclear Architecture: A Review of Chromatin Organization, Cohesins, and Condensins with a Focus on C. elegans
Abstract
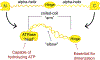
Chromatin is the complex of DNA and associated proteins found in the nuclei of living organisms. How it is organized is a major research field as it has implications for replication, repair, and gene expression. This review summarizes the current state of the chromatin organization field, with a special focus on chromatin motor complexes cohesin and condensin. Containing the highly conserved SMC proteins, these complexes are responsible for organizing chromatin during cell division. Additionally, research has demonstrated that condensin and cohesin also have important functions during interphase to shape the organization of chromatin and regulate expression of genes. Using the model organism C. elegans, the authors review the current knowledge of how these complexes perform such diverse roles and what open questions still exist in the field.
Keywords: C. elegans; SMC proteins; TADs; chromatin architecture; cohesin; condensin; dosage compensation.
Conflict of interest statement
Conflicts of Interest: The authors declare no competing financial interests.
Figures

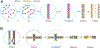

References
-
- Lee JY; Orr-Weaver TL Chromatin. In Encyclopedia of Genetics; Brenner S, Miller JH, Eds.; Academic Press: New York, NY, USA, 2001; pp. 340–343.
Grants and funding
LinkOut - more resources
Full Text Sources
