Pseudovibriamides from Pseudovibrio marine sponge bacteria promote flagellar motility via transcriptional modulation
- PMID: 39727420
- PMCID: PMC11796379
- DOI: 10.1128/mbio.03115-24
Pseudovibriamides from Pseudovibrio marine sponge bacteria promote flagellar motility via transcriptional modulation
Abstract
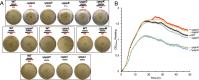
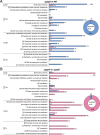
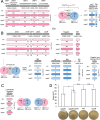
Pseudovibrio α-Proteobacteria have been repeatedly isolated from marine sponges and proposed to be beneficial to the host. Bacterial motility is known to contribute to host colonization. We have previously identified pseudovibriamides A and B, produced in culture by Pseudovibrio brasiliensis Ab134, and shown that pseudovibriamide A promotes flagellar motility. Pseudovibriamides are encoded in a hybrid nonribosomal peptide synthetase-polyketide synthase gene cluster that also includes several accessory genes. Pseudovibriamide A is a linear heptapeptide and pseudovibriamide B is a nonadepsipeptide derived from pseudovibriamide A. Here, we define the borders of the pseudovibriamides gene cluster, assign function to biosynthetic genes using reverse genetics, and test the hypothesis that pseudovibriamides impact motility by modulating gene transcription. RNA-sequencing transcriptomic analyses of strains having different compositions of pseudovibriamides suggested that both pseudovibriamides A and B affect genes potentially involved in motility, and that a compensatory mechanism is at play in mutants that produce only pseudovibriamide A, resulting in comparable flagellar motility as the wild type. The data gathered suggest that pseudovibriamides A and B have opposite roles in modulating a subset of genes, with pseudovibriamide B having a primary effect in gene activation, and pseudovibriamide A on inhibition. Finally, we observed many differentially expressed genes (up to 29% of the total gene number) indicating that pseudovibriamides have a global effect on transcription that goes beyond motility.IMPORTANCEMarine sponges are found throughout the oceans from tropical coral reefs to polar sea floors, playing crucial roles in marine ecosystems. Pseudovibrio bacteria have been proposed to contribute to sponge health. We have previously shown that pseudovibriamides produced by Pseudovibrio brasiliensis promote bacterial motility, a behavior that is beneficial to bacterial survival and host colonization. The gene cluster that encodes pseudovibriamide biosynthesis is found in two-thirds of Pseudovibrio genomes. This gene cluster is also present in Pseudomonas bacteria that interact with terrestrial plants and animals. Here, we first assign functions to pseudovibriamide biosynthetic genes using reverse genetics. We then show that pseudovibriamides play a major role in transcriptional regulation, affecting up to 29% of P. brasiliensis genes, including motility genes. Thus, this work gives insights into pseudovibriamide biosynthesis and provides evidence that they are signaling molecules relevant to bacterial motility and to other yet-to-be-identified phenotypes.
Keywords: flagellar motility; marine sponge; proteobacteria; secondary metabolite; transcriptomics.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2017 Dec 22;12(12):CD011535. doi: 10.1002/14651858.CD011535.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jan 9;1:CD011535. doi: 10.1002/14651858.CD011535.pub3. PMID: 29271481 Free PMC article. Updated.
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
Comparison of Two Modern Survival Prediction Tools, SORG-MLA and METSSS, in Patients With Symptomatic Long-bone Metastases Who Underwent Local Treatment With Surgery Followed by Radiotherapy and With Radiotherapy Alone.Clin Orthop Relat Res. 2024 Dec 1;482(12):2193-2208. doi: 10.1097/CORR.0000000000003185. Epub 2024 Jul 23. Clin Orthop Relat Res. 2024. PMID: 39051924
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2020 Jan 9;1(1):CD011535. doi: 10.1002/14651858.CD011535.pub3. Cochrane Database Syst Rev. 2020. Update in: Cochrane Database Syst Rev. 2021 Apr 19;4:CD011535. doi: 10.1002/14651858.CD011535.pub4. PMID: 31917873 Free PMC article. Updated.
Cited by
-
Evaluation of vectors for gene expression in Pseudovibrio marine bacteria.Appl Environ Microbiol. 2025 Apr 23;91(4):e0020725. doi: 10.1128/aem.00207-25. Epub 2025 Mar 4. Appl Environ Microbiol. 2025. PMID: 40035598 Free PMC article.
References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

