Canadian Consensus Recommendations for Predictive Biomarker Testing in Gastric and Gastroesophageal Junction Adenocarcinoma
- PMID: 39727695
- PMCID: PMC11674259
- DOI: 10.3390/curroncol31120572
Canadian Consensus Recommendations for Predictive Biomarker Testing in Gastric and Gastroesophageal Junction Adenocarcinoma
Abstract
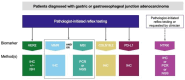
Gastric cancer is common globally and has a generally poor prognosis with a low 5-year survival rate. Targeted therapies and immunotherapies have improved the treatment landscape, providing more options for efficacious treatment. The use of these therapies requires predictive biomarker testing to identify patients who can benefit from their use. New therapies on the horizon, such as CLDN18.2 monoclonal antibody therapy, require laboratories to implement new biomarker tests. A multidisciplinary pan-Canadian expert working group was convened to develop guidance for pathologists and oncologists on the implementation of CLDN18.2 IHC testing for gastric and gastroesophageal junction (G/GEJ) adenocarcinoma in Canada, as well as general recommendations to optimize predictive biomarker testing in G/GEJ adenocarcinoma. The expert working group recommendations highlight the importance of reflex testing for HER2, MMR and/or MSI, CLDN18, and PD-L1 in all patients at first diagnosis of G/GEJ adenocarcinoma. Testing for NTRK fusions may also be included in reflex testing or requested by the treating clinician when third-line therapy is being considered. The expert working group also made recommendations for pre-analytic, analytic, and post-analytic considerations for predictive biomarker testing in G/GEJ adenocarcinoma. Implementation of these recommendations will provide medical oncologists with accurate, timely biomarker results to use for treatment decision-making.
Keywords: CLDN18; CLDN18.2; HER2; IHC; MMR; PD-L1; gastric cancer; immunohistochemistry; immunotherapy; targeted therapy.
Conflict of interest statement
All authors report honoraria from Astellas Canada for participation in working group meetings related to this project. Christine Brezden-Masley has received consultancy/advisory board honoraria from Astellas, AstraZeneca, BMS, Beigene, Eli Lilly, Gilead Sciences, Knight, Merck, Novartis, Pfizer, and Roche and research funding from Astellas, AstraZeneca, Eli Lilly, Pfizer, Novartis, and Gilead Sciences. Pierre O. Fiset has received consultancy/advisory boards/honoraria from Amgen, Astellas, AstraZeneca, EMD Serono, Roche, Incyte, Merck, Novartis, and Pfizer and research support and project funding from Astellas, AstraZeneca, Merck, Bristol Myers Squibb, Pfizer, CIHR-CRS, MGH Foundation, and Rossy Cancer Network. Thomas Arnason has received honoraria/consultancies/speakerships from Merck, Bristol Myers, and GSK. Diana N. Ionescu has received honoraria, grant support, and/or participated in advisory meetings with AstraZeneca, Pfizer, BMS, Roche, Merck, Amgen, Eli Lilly, and Bayer and has a leadership/fiduciary role with Lung Cancer Canada. Howard J Lim has received honoraria from BMS, Merck, and the Canadian Drug Agency. Brandon S. Sheffield has received honoraria, grant support, and/or participated in advisory meetings with Amgen, AstraZeneca, Bayer, Biocartis, Boehringer Ingelheim, Cell Marque, Elevation Oncology, Eli Lilly, EMD Serono, Incyte, Janssen, Merck, Novartis, Pfizer, Roche, Sanofi, ThermoFisher, and Turning Point Therapeutics. Catherine J. Streutker has received honoraria from Astellas for advisory board and speaker support, Roche and Merck for speaker support, and consulting fees from Abbvie and Boehringer Ingelheim.
Figures




Similar articles
-
Global prevalence of claudin 18 isoform 2 in tumors of patients with locally advanced unresectable or metastatic gastric or gastroesophageal junction adenocarcinoma.Gastric Cancer. 2024 Sep;27(5):1058-1068. doi: 10.1007/s10120-024-01518-1. Epub 2024 Jul 2. Gastric Cancer. 2024. PMID: 38954176 Free PMC article.
-
Immuno-Oncology Biomarkers for Gastric and Gastroesophageal Junction Adenocarcinoma: Why PD-L1 Testing May Not Be Enough.Oncologist. 2018 Oct;23(10):1171-1177. doi: 10.1634/theoncologist.2018-0034. Epub 2018 Apr 27. Oncologist. 2018. PMID: 29703766 Free PMC article.
-
Retrospective Study of Claudin 18 Isoform 2 Prevalence and Prognostic Association in Gastric and Gastroesophageal Junction Adenocarcinoma.JCO Precis Oncol. 2024 May;8:e2300543. doi: 10.1200/PO.23.00543. JCO Precis Oncol. 2024. PMID: 38781542 Free PMC article.
-
Claudin-18.2 Immunohistochemical Evaluation in Gastric and Gastroesophageal Junction Adenocarcinomas to Direct Targeted Therapy: A Practical Approach.Mod Pathol. 2024 Nov;37(11):100589. doi: 10.1016/j.modpat.2024.100589. Epub 2024 Aug 2. Mod Pathol. 2024. PMID: 39098518 Review.
-
Gastroesophageal Junction Adenocarcinoma: Is There an Optimal Management?Am Soc Clin Oncol Educ Book. 2019 Jan;39:e88-e95. doi: 10.1200/EDBK_236827. Epub 2019 May 17. Am Soc Clin Oncol Educ Book. 2019. PMID: 31099690 Review.
References
-
- World Cancer Research Fund International Stomach Cancer Statistics. [(accessed on 12 June 2024)]. Available online: https://www.wcrf.org/cancer-trends/stomach-cancer-statistics/
-
- Canadian Cancer Society Stomach Cancer Statistics. [(accessed on 12 June 2024)]. Available online: https://cancer.ca/en/cancer-information/cancer-types/stomach/statistics.
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

