Hybrid Chitosan Biosorbents: Tunable Adsorption at Surface and Micropore Domains
- PMID: 39727729
- PMCID: PMC11673820
- DOI: 10.3390/biomimetics9120725
Hybrid Chitosan Biosorbents: Tunable Adsorption at Surface and Micropore Domains
Abstract
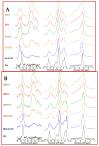
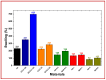
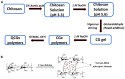
Herein, we report a study that provides new insight on the knowledge gaps that relate to the role of biopolymer structure and adsorption properties for chitosan adsorbents that are cross-linked with glutaraldehyde. The systematic modification of chitosan cross-linked with glutaraldehyde (CG) and its quaternized forms (QCG) was studied in relation to the reaction conditions: mole ratios of reactants and pH conditions. Complementary adsorbent characterization employed 13C NMR/FTIR spectroscopy, TGA and DSC, point-zero-charge (PZC), solvent swelling, and sorption studies using selected dye probes. The spectral and thermal techniques provide complementary evidence that affirm the key role of cross-linker content and quaternization on variation of the physicochemical properties of chitosan. The PZC results reveal a neutral surface charge for the modified materials between pH 6.0 to 6.3 ± 0.3, as compared with pH 8.7 ± 0.4 for pristine chitosan. Solvent swelling in water decreased with greater cross-linking, while the QCG materials had greater swelling over CG materials due to enhanced hydration. The adsorption results reveal variable dye uptake properties according to the cross-linker content. Similarly, surface versus micropore adsorption was demonstrated, according to the nature and ionization state of the dye for the modified adsorbents, where the CG and QCG materials had tunable sorption properties that exceeded that of unmodified chitosan. A key step in tuning the structure and surface chemical properties of cross-linked chitosan involves pH control during synthesis. The facile tunability of the physicochemical properties of the modified biopolymers reported herein means that they possess features of biomimetics that are relevant to advanced drug delivery, antimicrobial materials for wound healing, biosensors, and biosorbents for biomedical applications.
Keywords: adsorption; biopolymer cross-linking; chitosan; glutaraldehyde; pore structure; surface quaternization.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures





References
-
- Roberts G.A.F. Structure of Chitin and Chitosan. In: Roberts G.A.F., editor. Chitin Chemistry. Macmillan Education UK; London, UK: 1992. pp. 1–53. - DOI
-
- Li Q., Dunn E.T., Grandmaison E.W., Goosen M.F.A. Applications and Properties of Chitosan. J. Bioact. Compat. Polym. 1992;7:370–397. doi: 10.1177/088391159200700406. - DOI
-
- Alqahtani N.F. Functionalized imidazolium ionic liquids-modified chitosan materials: From synthesis approaches to applications. React. Funct. Polym. 2024;194:105779. doi: 10.1016/j.reactfunctpolym.2023.105779. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

