Therapeutic Potential of Oligo-Fucoidan in Mitigating Peritoneal Dialysis-Associated Fibrosis
- PMID: 39728104
- PMCID: PMC11678284
- DOI: 10.3390/md22120529
Therapeutic Potential of Oligo-Fucoidan in Mitigating Peritoneal Dialysis-Associated Fibrosis
Abstract
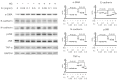
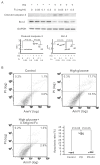
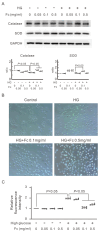
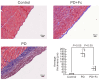
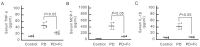
Peritoneal dialysis (PD) serves as a home-based kidney replacement therapy with increasing utilization across the globe. However, long-term use of high-glucose-based PD solution incites repeated peritoneal injury and inevitable peritoneal fibrosis, thus compromising treatment efficacy and resulting in ultrafiltration failure eventually. In the present study, we utilized human mesothelial MeT-5A cells for the in vitro experiments and a PD mouse model for in vivo validation to study the pathophysiological mechanisms underneath PD-associated peritoneal fibrosis. High-glucose PD solution (Dianeal 4.25%, Baxter) increased protein expression of mesothelial-mesenchymal transition (MMT) markers, such as N-cadherin and α-SMA in MeT-5A cells, whereas it decreased catalase expression and stimulated the production of reactive oxygen species (ROS). Furthermore, macrophage influx and increased serum pro-inflammatory cytokines, such as IL-1β, MCP-1, and TNF-α, were observed in the PD mouse model. Interestingly, we discovered that oligo-fucoidan, an oligosaccharide extract from brown seaweed, successfully prevented PD-associated peritoneal thickening and fibrosis through antioxidant effect, downregulation of MMT markers, and attenuation of peritoneal and systemic inflammation. Hence, oligo-fucoidan has the potential to be developed into a novel preventive strategy for PD-associated peritoneal fibrosis.
Keywords: MMT; fibrosis; mesothelial–mesenchymal transition; oligo-fucoidan; peritoneal dialysis.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures

References
-
- Raoofi S., Pashazadeh Kan F., Rafiei S., Hoseinipalangi Z., Rezaei S., Ahmadi S., Masoumi M., Noorani Mejareh Z., Roohravan Benis M., Sharifi A., et al. Hemodialysis and peritoneal dialysis-health-related quality of life: Systematic review plus meta-analysis. BMJ Support. Palliat. Care. 2023;13:365–373. doi: 10.1136/bmjspcare-2021-003182. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

