SLAMF7 defines subsets of human effector CD8 T cells
- PMID: 39730488
- PMCID: PMC11680708
- DOI: 10.1038/s41598-024-80971-5
SLAMF7 defines subsets of human effector CD8 T cells
Abstract
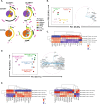
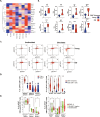
Long-term control of viral replication relies on the efficient differentiation of memory T cells into effector T cells during secondary immune responses. Recent findings have identified T cell precursors for both memory and exhausted T cells, suggesting the existence of progenitor-like effector T cells. These cells can persist without antigenic challenge but expand and acquire effector functions upon recall immune responses. In this study, we demonstrate that the combination of SLAMF7 with either CD27 or TCF-1 effectively identifies progenitor-like effector CD8 T cells, while SLAMF7 with GPR56 or TOX defines effector CD8 T cells. These markers allow for the clear segregation of these distinct cell subsets. SLAMF7+ CD8T cells are dynamically modulated during viral infections, including HIV, HCV, CMV, and SARS-CoV-2, as well as during aging. We further characterize the SLAMF7 signature at both phenotypic and transcriptional levels. Notably, during aging, the SLAMF7 pathway becomes dysregulated, resulting in persistent phosphorylation of STAT1. Additionally, SLAMF7 ligation in the presence of IL-15 induces TCF-1 expression, which promotes the homeostatic proliferation of progenitor-like effector CD8 T cells.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests.
Figures



References
MeSH terms
Substances
Grants and funding
- No. 10-036/Agency for Science, Technology and Research (A*STAR)
- BMRC IAF 311006, BMRC transition funds#H16/99/b0/011, BMRC IAF-PP H1901a0024/Agency for Science, Technology and Research (A*STAR)
- PI16/00684/Ministry of Economy and Competitiveness | Instituto de Salud Carlos III (Institute of Health Carlos III)
- PI9/01127/Ministry of Economy and Competitiveness | Instituto de Salud Carlos III (Institute of Health Carlos III)
- HIR/MOHE; H-20001-E000001/Ministry of Higher Education, Malaysia (MOHE)
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

