A Neutrophil Extracellular Traps-Related Signature Predicts Clinical Outcomes and Identifies Immune Landscape in Ovarian Cancer
- PMID: 39730971
- PMCID: PMC11680186
- DOI: 10.1111/jcmm.70302
A Neutrophil Extracellular Traps-Related Signature Predicts Clinical Outcomes and Identifies Immune Landscape in Ovarian Cancer
Abstract
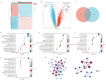
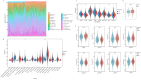
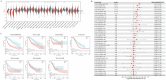
Ovarian cancer (OvCa) is the most lethal gynaecology malignancies worldwide. Neutrophil extracellular traps (NETs), net-like protein structures produced by activated neutrophils and DNA-histone complexes, have a central role in tumours, though haven't been fully explored in OvCa. We obtained transcriptome data from TCGA-OvCa database (n = 376) as training, ICGC-OvCa database (n = 111) as validation and GTEx database (n = 180) as controls. Through LASSO-COX Regression analysis, we identified an eight-gene signature among 87 NETs-related genes, which was significantly related to poor prognosis in both TCGA-OvCa and ICGC-OvCa cohorts (Log-rank p-value = 0.0003 and 0.0014). Next, we constructed and validated a prognostic nomogram, consist of NETs-related signature and clinical features (C-index = 0.82). We evaluated 22 typical immune cell infiltration through CIBERSORT analysis, which implied upregulation of memory CD4 + T cells, follicular helper T cells and neutrophils in high-risk group. Additionally, we predicted therapy sensitivity through TIDE algorithm, indicating that high NETs-riskscore exhibited more sensitivity towards Sorafenib and less sensitivity towards immunotherapy. We initially reported that RAC2 upregulation was associated with NETs formation and poor prognosis (p-value < 0.05) through IHC analysis of tissue microarrays (n = 125). Conclusively, NETs-related signature was reliable for OvCa prognosis prediction and therapy assessment. Especially, RAC2 was predominantly related to NETs formation, thus providing hints towards anti-tumour mechanism of NETs in OvCa.
Keywords: RAC2; neutrophil extracellular traps; ovarian cancer; prognostic signature; tumour immune landscape.
© 2024 The Author(s). Journal of Cellular and Molecular Medicine published by Foundation for Cellular and Molecular Medicine and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

