Choline metabolism modulates cyclic-di-GMP signaling and virulence of Pseudomonas aeruginosa in a macrophage infection model
- PMID: 39731097
- PMCID: PMC11681757
- DOI: 10.1186/s12879-024-10375-3
Choline metabolism modulates cyclic-di-GMP signaling and virulence of Pseudomonas aeruginosa in a macrophage infection model
Abstract
Background: Bacterial pathogens frequently encounter host-derived metabolites during their colonization and invasion processes, which can serve as nutrients, antimicrobial agents, or signaling molecules for the pathogens. The essential nutrient choline (Cho) is widely known to be utilized by a diverse range of bacteria and may undergo conversion into the disease-associated metabolite trimethylamine (TMA). However, the impact of choline metabolism on bacterial physiology and virulence remains largely unexplored.
Methods: Here, we employed an in vitro infection model to investigate the role of Cho in intracellular survival and virulence of Pseudomonas aeruginosa (P. aeruginosa). Additionally, a comprehensive RNA-seq based transcriptomic analysis and various phenotypic assays were performed to elucidate the impacts of Cho on P. aeruginosa.
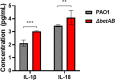
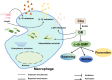
Results: We observed that the Cho metabolite glycine betaine (GB) effectively reduced intracellular levels of cyclic-di-GMP (c-di-GMP). Supplementation of Cho or GB in P. aeruginosa had thus affected c-di-GMP regulated phenotypes, such as pyoverdine production, biofilm formation, and mobility. Depletion of Cho metabolism through knockout of the betAB operon resulted in compromised intracellular survival of P. aeruginosa. Notably, the P. aeruginosa betAB mutant elicited a more robust protective inflammatory response compared to the wild-type strain.
Conclusion: Our study showed that P. aeruginosa Cho metabolism not only interferes host nutritional immunity, but also directly affect multiple virulence phenotypes through modulation of c-di-GMP signaling.
Keywords: Pseudomonas aeruginosa; Biofilm; Choline metabolism; Cyclic-di-GMP; Virulence.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures

Similar articles
-
Reduced Intracellular c-di-GMP Content Increases Expression of Quorum Sensing-Regulated Genes in Pseudomonas aeruginosa.Front Cell Infect Microbiol. 2017 Oct 17;7:451. doi: 10.3389/fcimb.2017.00451. eCollection 2017. Front Cell Infect Microbiol. 2017. PMID: 29090193 Free PMC article.
-
Cyclic Di-GMP Signaling Contributes to Pseudomonas aeruginosa-Mediated Catheter-Associated Urinary Tract Infection.J Bacteriol. 2015 Jul 20;198(1):91-7. doi: 10.1128/JB.00410-15. Print 2016 Jan 1. J Bacteriol. 2015. PMID: 26195591 Free PMC article.
-
The Cyclic AMP-Vfr Signaling Pathway in Pseudomonas aeruginosa Is Inhibited by Cyclic Di-GMP.J Bacteriol. 2015 Jul;197(13):2190-200. doi: 10.1128/JB.00193-15. Epub 2015 Apr 20. J Bacteriol. 2015. PMID: 25897033 Free PMC article.
-
Biofilms and Cyclic di-GMP (c-di-GMP) Signaling: Lessons from Pseudomonas aeruginosa and Other Bacteria.J Biol Chem. 2016 Jun 10;291(24):12547-12555. doi: 10.1074/jbc.R115.711507. Epub 2016 Apr 21. J Biol Chem. 2016. PMID: 27129226 Free PMC article. Review.
-
c-di-GMP and its Effects on Biofilm Formation and Dispersion: a Pseudomonas Aeruginosa Review.Microbiol Spectr. 2015 Apr;3(2):MB-0003-2014. doi: 10.1128/microbiolspec.MB-0003-2014. Microbiol Spectr. 2015. PMID: 26104694 Free PMC article. Review.
Cited by
-
Cross-membrane cooperation among bacteria can facilitate intracellular pathogenesis.Nat Commun. 2025 Aug 11;16(1):7419. doi: 10.1038/s41467-025-62575-3. Nat Commun. 2025. PMID: 40790115 Free PMC article.
References
-
- Whiteley M. The host as a growth medium: targeting bacterial nutrient uptake in New Treatment Strategies. Pediatr Pulmonol. 2013;48:154–55.
-
- Chittim CL, del Campo AM, Balskus EP. Gut bacterial phospholipase ds support disease-associated metabolism by generating choline. Nat Microbiol. 2019;4:155–63. - PubMed
-
- Kortstee GJ. The aerobic decomposition of choline by microorganisms. I. The ability of aerobic organisms, particularly coryneform bacteria, to utilize choline as the sole carbon and nitrogen source. Archives Microbiol. 1970;71:235–44. - PubMed
-
- Lisa TA, Garrido MN, Domenech CE. Induction of acid phosphatase and cholinesterase activities in ps. Aeruginosa and their in-vitro control by choline, acetylcholine and betaine. Mol Cell Biochem. 1983;50:149–55. - PubMed
MeSH terms
Substances
Grants and funding
- 2024-HY013/HaiYa Young Scientist Foundation of Shenzhen University General Hospital
- KQTD20200909113758004/Shenzhen Science and Technology Program
- 91951204/National Natural Science Foundation of China
- 2019QN01Y163/Guangdong High-level Personnel of Special Support Program
- 2019A1515110640/Guangdong Basic and Applied Basic Research Foundation
LinkOut - more resources
Full Text Sources

