Tppp3 is a novel molecule for retinal ganglion cell identification and optic nerve regeneration
- PMID: 39734233
- PMCID: PMC11684310
- DOI: 10.1186/s40478-024-01917-6
Tppp3 is a novel molecule for retinal ganglion cell identification and optic nerve regeneration
Abstract
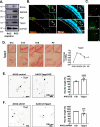
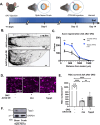
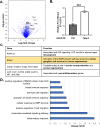
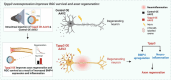
Mammalian central nervous system (CNS) axons cannot spontaneously regenerate after injury, creating an unmet need to identify molecular regulators to promote axon regeneration and reduce the lasting impact of CNS injuries. While tubulin polymerization promoting protein family member 3 (Tppp3) is known to promote axon outgrowth in amphibians, its role in mammalian axon regeneration remains unknown. Here we investigated Tppp3 in retinal ganglion cells (RGCs) neuroprotection and axonal regeneration using an optic nerve crush (ONC) model in the rodent. Single-cell RNA sequencing identified the expression of Tppp3 in RGCs of mice, macaques, and humans. Tppp3 overexpression enhanced neurite outgrowth in mouse primary RGCs in vitro, promoted axon regeneration, and improved RGC survival after ONC. Bulk RNA sequencing indicated that Tppp3 overexpression upregulates axon regeneration genes such as Bmp4 and neuroinflammatory pathways. Our findings advance regenerative medicine by developing a new therapeutic strategy for RGC neuroprotection and axon regeneration.
Keywords: Axon regeneration; BMP4; Inflammation; Neurite outgrowth; Retinal ganglion cells; Tppp3.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: The study was conducted in compliance with the ARVO guidelines and approved by IACUC at the University of Pittsburgh. Consent for publication: Not applicable. Competing interests: KCC and JLG are co-inventors on a patent application submitted through Stanford University. The authors declare no other competing interests.
Figures

Similar articles
-
Tumor Necrosis Factor Receptor Superfamily Member 12A Enhances Retinal Ganglion Cell Survival and Promotes Axon Regeneration.FASEB J. 2025 Aug 15;39(15):e70895. doi: 10.1096/fj.202500988RR. FASEB J. 2025. PMID: 40747811
-
Interleukin-4 protects retinal ganglion cells and promotes axon regeneration.Cell Commun Signal. 2024 Apr 22;22(1):236. doi: 10.1186/s12964-024-01604-y. Cell Commun Signal. 2024. PMID: 38650003 Free PMC article.
-
Co-delivery of neurotrophic factors and a zinc chelator substantially increases retinal ganglion cell survival and axon protection in the optic nerve crush model.Acta Biomater. 2025 Jul 1;201:297-308. doi: 10.1016/j.actbio.2025.06.007. Epub 2025 Jun 5. Acta Biomater. 2025. PMID: 40482982 Free PMC article.
-
Neuroinflammation, Microglia and Implications for Retinal Ganglion Cell Survival and Axon Regeneration in Traumatic Optic Neuropathy.Front Immunol. 2022 Mar 4;13:860070. doi: 10.3389/fimmu.2022.860070. eCollection 2022. Front Immunol. 2022. PMID: 35309305 Free PMC article. Review.
-
Emerging therapeutic strategies for optic nerve regeneration.Trends Pharmacol Sci. 2025 Jan;46(1):45-61. doi: 10.1016/j.tips.2024.11.008. Epub 2024 Dec 17. Trends Pharmacol Sci. 2025. PMID: 39694789 Review.
Cited by
-
Advancing Parkinson's diagnosis: seed amplification assay for α-synuclein detection in minimally invasive samples.Mol Cell Biochem. 2025 Jun;480(6):3297-3314. doi: 10.1007/s11010-024-05190-y. Epub 2025 Jan 6. Mol Cell Biochem. 2025. PMID: 39760833 Review.
References
-
- Aoki M, Segawa H, Naito M, Okamoto H (2014) Identification of possible downstream genes required for the extension of peripheral axons in primary sensory neurons. Biochem Biophys Res Commun 445:357–362. 10.1016/j.bbrc.2014.01.193 - PubMed
-
- Barnstable CJ, Drager UC (1984) Thy-1 antigen: a ganglion cell specific marker in rodent retina. Neuroscience 11:847–855. 10.1016/0306-4522(84)90195-7 - PubMed
-
- Barres BA, Silverstein BE, Corey DP, Chun LL (1988) Immunological, morphological, and electrophysiological variation among retinal ganglion cells purified by panning. Neuron 1:791–803. 10.1016/0896-6273(88)90127-4 - PubMed
-
- Benowitz LI, He Z, Goldberg JL (2017) Reaching the brain: advances in optic nerve regeneration. Exp Neurol 287:365–373. 10.1016/j.expneurol.2015.12.015 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

