Molecular and metabolic landscape of adenosine triphosphate-induced cell death in cardiovascular disease
- PMID: 39734818
- PMCID: PMC11669974
- DOI: 10.4330/wjc.v16.i12.689
Molecular and metabolic landscape of adenosine triphosphate-induced cell death in cardiovascular disease
Abstract
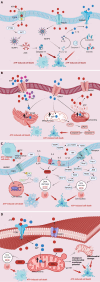
The maintenance of intracellular and extracellular adenosine triphosphate (ATP) levels plays a pivotal role in cardiac function. In recent years, burgeoning attention has been directed towards ATP-induced cell death (AICD), revealing it as a distinct cellular demise pathway triggered by heightened extracellular ATP concentrations, distinguishing it from other forms of cell death such as apoptosis and necrosis. AICD is increasingly acknowledged as a critical mechanism mediating the pathogenesis and progression of various cardiovascular maladies, encompassing myocardial ischemia-reperfusion injury, sepsis-induced cardiomyopathy, hypertrophic cardiomyopathy, arrhythmia, and diabetic cardiomyopathy. Consequently, a comprehensive understanding of the molecular and metabolic underpinnings of AICD in cardiac tissue holds promise for the prevention and amelioration of cardiovascular diseases. This review first elucidates the vital physiological roles of ATP in the cardiovascular system, subsequently delving into the intricate molecular mechanisms and metabolic signatures governing AICD. Furthermore, it addresses the potential therapeutic targets implicated in mitigating AICD for treating cardiovascular diseases, while also delineating the current constraints and future avenues for these innovative therapeutic targets, thereby furnishing novel insights and strategies for the prevention and management of cardiovascular disorders.
Keywords: Adenosine triphosphate induced cell death; Cardiovascular diseases; Metabolic pathways; Molecular mechanisms; Myocardial ischemia-reperfusion injury.
©The Author(s) 2024. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: The authors report no relevant conflicts of interest for this article.
Figures
References
-
- Galluzzi L, Vitale I, Aaronson SA, Abrams JM, Adam D, Agostinis P, Alnemri ES, Altucci L, Amelio I, Andrews DW, Annicchiarico-Petruzzelli M, Antonov AV, Arama E, Baehrecke EH, Barlev NA, Bazan NG, Bernassola F, Bertrand MJM, Bianchi K, Blagosklonny MV, Blomgren K, Borner C, Boya P, Brenner C, Campanella M, Candi E, Carmona-Gutierrez D, Cecconi F, Chan FK, Chandel NS, Cheng EH, Chipuk JE, Cidlowski JA, Ciechanover A, Cohen GM, Conrad M, Cubillos-Ruiz JR, Czabotar PE, D'Angiolella V, Dawson TM, Dawson VL, De Laurenzi V, De Maria R, Debatin KM, DeBerardinis RJ, Deshmukh M, Di Daniele N, Di Virgilio F, Dixit VM, Dixon SJ, Duckett CS, Dynlacht BD, El-Deiry WS, Elrod JW, Fimia GM, Fulda S, García-Sáez AJ, Garg AD, Garrido C, Gavathiotis E, Golstein P, Gottlieb E, Green DR, Greene LA, Gronemeyer H, Gross A, Hajnoczky G, Hardwick JM, Harris IS, Hengartner MO, Hetz C, Ichijo H, Jäättelä M, Joseph B, Jost PJ, Juin PP, Kaiser WJ, Karin M, Kaufmann T, Kepp O, Kimchi A, Kitsis RN, Klionsky DJ, Knight RA, Kumar S, Lee SW, Lemasters JJ, Levine B, Linkermann A, Lipton SA, Lockshin RA, López-Otín C, Lowe SW, Luedde T, Lugli E, MacFarlane M, Madeo F, Malewicz M, Malorni W, Manic G, Marine JC, Martin SJ, Martinou JC, Medema JP, Mehlen P, Meier P, Melino S, Miao EA, Molkentin JD, Moll UM, Muñoz-Pinedo C, Nagata S, Nuñez G, Oberst A, Oren M, Overholtzer M, Pagano M, Panaretakis T, Pasparakis M, Penninger JM, Pereira DM, Pervaiz S, Peter ME, Piacentini M, Pinton P, Prehn JHM, Puthalakath H, Rabinovich GA, Rehm M, Rizzuto R, Rodrigues CMP, Rubinsztein DC, Rudel T, Ryan KM, Sayan E, Scorrano L, Shao F, Shi Y, Silke J, Simon HU, Sistigu A, Stockwell BR, Strasser A, Szabadkai G, Tait SWG, Tang D, Tavernarakis N, Thorburn A, Tsujimoto Y, Turk B, Vanden Berghe T, Vandenabeele P, Vander Heiden MG, Villunger A, Virgin HW, Vousden KH, Vucic D, Wagner EF, Walczak H, Wallach D, Wang Y, Wells JA, Wood W, Yuan J, Zakeri Z, Zhivotovsky B, Zitvogel L, Melino G, Kroemer G. Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 2018;25:486–541. - PMC - PubMed
-
- Zhang HL, Sandai D, Zhang ZW, Song ZJ, Babu D, Tabana Y, Dahham SS, Adam Ahmed Adam M, Wang Y, Wang W, Zhang HL, Zhao R, Barakat K, Harun MSR, Shapudin SNM, Lok B. Adenosine triphosphate induced cell death: Mechanisms and implications in cancer biology and therapy. World J Clin Oncol. 2023;14:549–569. - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

