Proportional Assist Ventilation for Minimizing the Duration of Mechanical Ventilation (the PROMIZING study): update to the statistical analysis plan for a randomized controlled trial
- PMID: 39736673
- PMCID: PMC11684088
- DOI: 10.1186/s13063-024-08669-7
Proportional Assist Ventilation for Minimizing the Duration of Mechanical Ventilation (the PROMIZING study): update to the statistical analysis plan for a randomized controlled trial
Abstract
Background: We previously published the protocol and statistical analysis plan for a randomized controlled trial of Proportional Assist Ventilation for Minimizing the Duration of Mechanical Ventilation: the PROMIZING study in Trials ( https://doi.org/10.1186/s13063-023-07163-w ). This update summarizes changes made to the statistical analysis plan for the trial since the publication of the original protocol and statistical analysis plan.
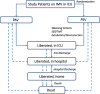
Methods/design: The Proportional Assist Ventilation for Minimizing the Duration of Mechanical Ventilation (PROMIZING) study is a multi-center, open-label, randomized controlled trial designed to determine if ventilation with proportional assist ventilation with load-adjustable gain factors will result in a shorter duration of time spent on mechanical ventilation compared to ventilation with pressure support ventilation for patients with acute respiratory failure. The statistical analysis plan for the trial was incorporated into the original publication of the protocol in Trials ( https://doi.org/10.1186/s13063-023-07163-w ) and was based on version 5.0 of the study protocol and version 1.0 of the statistical analysis plan (SAP), which included plans for both frequentist and Bayesian analyses. We have since updated the SAP to refine the Bayesian analysis plan, update the multistate model diagram, and include plans for a cluster analysis to determine if there is heterogeneity of treatment effect. This update summarizes the changes made and their rationale and provides a refined SAP for the PROMIZING trial with additional background information, in adherence with guidelines for the prospective reporting of SAPs for randomized controlled trials.
Trial registration: ClinicalTrials.gov Identifier: NCT02447692 prospectively registered May 19, 2015.
Keywords: Bayesian; Cluster; Liberation; Proportional assist ventilation; Statistical analysis plan; Ventilator-free days; Weaning from mechanical ventilation.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Ethics approval was granted from the Western University Research Ethics Board through Clinical Trials Ontario (CTO #0733) for the PROMIZING study. Written, informed consent to participate will be obtained from all participants or their Substitute Decision Maker (SDM). All centers participating in the study get their own ethics committee approval. Consent for publication: Not applicable. Competing interests: The authors declare they have no direct competing interests in the study. The study received partial industry funding through the first grant which was a CIHR-industry partnership, with the industry partner at arm’s length. The industry partner, Medtronic, has no role in the study design, data collection, analysis, interpretation, or publication of manuscript. No author received personal financial remuneration for the study. The authors declare no other competing interests in the study.
Figures
References
-
- Carteaux G, Mancebo J, Mercat A, Dellamonica J, Richard JC, Aguirre-Bermeo H, Kouatchet A, Beduneau G, Thille AW, Brochard L. Bedside adjustment of proportional assist ventilation to target a predefined range of respiratory effort. Crit Care Med. 2013;41(9):2125–32. 10.1097/CCM.0b013e31828a42e5. PMID: 23787397. - DOI - PubMed
-
- Gamble C, Krishan A, Stocken D, Lewis S, Juszczak E, Doré C, Williamson PR, Altman DG, Montgomery A, Lim P, Berlin J, Senn S, Day S, Barbachano Y, Loder E. Guidelines for the content of statistical analysis plans in clinical trials. JAMA. 2017;318(23):2337–43. 10.1001/jama.2017.18556. PubMed PMID: 29260229. - DOI - PubMed
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous