Fatal outcome related to drug reaction with eosinophilia and systemic symptoms: a disproportionality analysis of FAERS database and a systematic review of cases
- PMID: 39737180
- PMCID: PMC11683082
- DOI: 10.3389/fimmu.2024.1490334
Fatal outcome related to drug reaction with eosinophilia and systemic symptoms: a disproportionality analysis of FAERS database and a systematic review of cases
Abstract
Background: Drug rash with eosinophilia and systemic symptoms (DRESS) is a life-threatening severe cutaneous adverse reaction.
Objective: This study aims to study fatal DRESS cases using FAERS database and systematic review.
Methods: Data of the FDA Adverse Event Reporting System (FAERS) database were extracted and manipulated. Articles from Pubmed, Embase and CINAHL databases were screened.
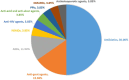
Results: 0.13% of the adverse events submitted to FAERS was identified as DRESS and the percentage of fatal cases was up to 6.62%. The top five drugs calculated to induce DRESS with the highest number of reported cases were allopurinol, lamotrigine, vancomycin, amoxicillin and carbamazepine. The top five drugs statistically related to fatal outcome with the highest number of reported cases were allopurinol, vancomycin, trimethoprim, sulfamethoxazole and lamotrigine. Skin manifestations remained the main reason for admission and the average time from dose to rash onset was 27.19 days. The most commonly cited culprit medication type were antibiotics (50.00%), anti-gout agents (15.38%) and anti-epileptic drug (11.54%).
Conclusions: We discussed fatal cases of DRESS through FAERS system and case reports, hoping to raise awareness when using relevant drugs.
Keywords: FDA adverse event reporting system; disproportionality analysis; drug adverse reaction; drug reaction with eosinophilia and systemic symptoms; drug-induced hypersensitivity syndrome; fatal cases; systematic review.
Copyright © 2024 Liang, An, Zhang, Liu and Zhang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Wei BM, Fox LP, Kaffenberger BH, Korman AM, Micheletti RG, Mostaghimi A, et al. Drug-induced hypersensitivity syndrome / drug reaction with eosinophilia and systemic symptoms. Part I. Epidemiology, pathogenesis, clinicopathological features, and prognosis. J Am Acad Dermatol. (2024) 90(5):885–908. doi: 10.1016/j.jaad.2023.02.072 - DOI - PubMed
-
- Kardaun SH, Sidoroff A, Valeyrie-Allanore L, Halevy S, Davidovici BB, Mockenhaupt M, et al. Variability in the clinical pattern of cutaneous side-effects of drugs with systemic symptoms: does a DRESS syndrome really exist? Br J Dermatol. (2007) 156:609–11. doi: 10.1111/j.1365-2133.2006.07704.x - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources