Longitudinal network changes and phenoconversion risk in isolated REM sleep behavior disorder
- PMID: 39737936
- PMCID: PMC11686016
- DOI: 10.1038/s41467-024-54695-z
Longitudinal network changes and phenoconversion risk in isolated REM sleep behavior disorder
Abstract
Isolated rapid eye movement sleep behavior disorder is a prodrome of α-synucleinopathies. Using positron emission tomography, we assessed changes in Parkinson's disease-related motor and cognitive metabolic networks and caudate/putamen dopaminergic input in a 4-year longitudinal imaging study of 13 male subjects with this disorder. We also correlated times to phenoconversion with baseline network expression in an independent validation sample. Expression values of both Parkinson's disease-related networks increased over time while dopaminergic input gradually declined in the longitudinal cohort. While abnormal functional connections were identified at baseline in both networks, others bridging these networks appeared later. These changes resulted in compromised information flow through the networks years before phenoconversion. We noted an inverse correlation between baseline network expression and times to phenoconversion to Parkinson's disease or dementia with Lewy bodies in the validation sample. Here, we show that the rate of network progression is a useful outcome measure in disease modification trials.
© 2024. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures



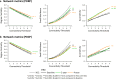
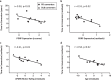

Update of
-
Longitudinal Network Changes and Phenoconversion Risk in Isolated REM Sleep Behavior Disorder.Res Sq [Preprint]. 2024 May 28:rs.3.rs-4427198. doi: 10.21203/rs.3.rs-4427198/v1. Res Sq. 2024. Update in: Nat Commun. 2024 Dec 30;15(1):10797. doi: 10.1038/s41467-024-54695-z. PMID: 38853923 Free PMC article. Updated. Preprint.
References
-
- Berg, D. et al. MDS research criteria for prodromal Parkinson’s disease. Mov. Disord.30, 1600–1611 (2015). - PubMed
-
- Galbiati, A., Verga, L., Giora, E., Zucconi, M. & Ferini-Strambi, L. The risk of neurodegeneration in REM sleep behavior disorder: A systematic review and meta-analysis of longitudinal studies. Sleep. Med. Rev.43, 37–46 (2019). - PubMed
-
- Schindlbeck, K. A. & Eidelberg, D. Network imaging biomarkers: insights and clinical applications in Parkinson’s disease. Lancet Neurol.17, 629–640 (2018). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

