A single mutation in dairy cow-associated H5N1 viruses increases receptor binding breadth
- PMID: 39737954
- PMCID: PMC11685663
- DOI: 10.1038/s41467-024-54934-3
A single mutation in dairy cow-associated H5N1 viruses increases receptor binding breadth
Abstract
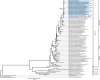
Clade 2.3.4.4b H5N1 is causing an unprecedented outbreak in dairy cows in the United States. To understand if recent H5N1 viruses are changing their receptor use, we screened recombinant hemagglutinin (HA) from historical and recent 2.3.4.4b H5N1 viruses for binding to distinct glycans bearing terminal sialic acids using a glycan microarray. We find that H5 from A/Texas/37/2024, an isolate from the dairy cow outbreak, has increased binding breadth to core glycans bearing terminal α2,3 sialic acids, the avian receptor, compared to historical and recent 2.3.4.4b H5N1 viruses. We do not observe any binding to α2,6 sialic acids, the receptor used by human seasonal influenza viruses. Using molecular dynamics and a cryo-EM structure of A/Texas/37/2024 H5, we show A/Texas/37/2024 H5 is more flexible within the receptor-binding site compared to a 2.3.4.4b H5 from 2022. We identify a single mutation outside of the receptor binding site, T199I, is responsible for increased binding breadth, as it increases receptor binding site flexibility. Together, these data show recent H5N1 viruses are evolving increased receptor binding breadth which could impact the host range and cell types infected with H5N1.
© 2024. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures



References
-
- USDA. Highly Pathogenic Avian Influenza (HPAI) Detections in Livestock, https://www.aphis.usda.gov/livestock-poultry-disease/avian/avian-influen... (2024).
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

