The interdependent hemodynamic influence between abdominal aortic aneurysm and renal artery stenosis
- PMID: 39738423
- PMCID: PMC11685789
- DOI: 10.1038/s41598-024-83622-x
The interdependent hemodynamic influence between abdominal aortic aneurysm and renal artery stenosis
Abstract
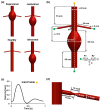
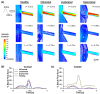
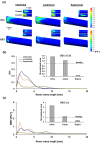
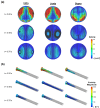
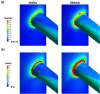
Cardiovascular diseases remain a leading cause of morbidity and mortality worldwide with abdominal aortic aneurysm (AAA) and renal artery stenosis (RAS) standing out as significant contributors to the vascular pathology spectrum. While these conditions have traditionally been approached as distinct entities, emerging evidence suggests a compelling interdependent relationship between AAA and RAS, challenging the conventional siloed understanding. The confluence of AAA and RAS represents a complex interplay within the cardiovascular system, one that is often overlooked in clinical practice and research. Here, we reveal a bidirectional consequential impact between these two diseases. The location of the AAA sac was investigated for its specific influence on the risk of RAS development. Although studies have shown a higher coincidence between the suprarenal AAA and RAS, our findings demonstrated that the presence of a suprarenal AAA correlated with the lowest risk of RAS development among the three investigated AAA locations. Notably, we also highlighted that the pre-existence of stenosis in the renal artery poses an elevated risk for the formation of suprarenal AAA, assessed by an increased wall shear stress gradient on the aortic wall. Our findings prompt a paradigm shift in the understanding and treatment of AAA and RAS in clinical practice.
© 2024. The Author(s).
Conflict of interest statement
Competing Interests: The author(s) have declared no conflicts of interest, financial or otherwise.
Figures
Similar articles
-
Does lower limb exercise worsen renal artery hemodynamics in patients with abdominal aortic aneurysm?PLoS One. 2015 May 6;10(5):e0125121. doi: 10.1371/journal.pone.0125121. eCollection 2015. PLoS One. 2015. PMID: 25946196 Free PMC article.
-
Abdominal aortic aneurysm and renal artery stenosis: renal function and blood pressure before and after endovascular treatment.J Hum Hypertens. 2002 May;16(5):367-9. doi: 10.1038/sj.jhh.1001367. J Hum Hypertens. 2002. PMID: 12082500
-
Combined renal artery stenosis and aortic aneurysm: treatment options.Ann Vasc Surg. 1996 Jul;10(4):361-4. doi: 10.1007/BF02286780. Ann Vasc Surg. 1996. PMID: 8879391
-
How does hemodynamics affect rupture tissue mechanics in abdominal aortic aneurysm: Focus on wall shear stress derived parameters, time-averaged wall shear stress, oscillatory shear index, endothelial cell activation potential, and relative residence time.Comput Biol Med. 2023 Mar;154:106609. doi: 10.1016/j.compbiomed.2023.106609. Epub 2023 Jan 23. Comput Biol Med. 2023. PMID: 36724610 Review.
-
Biomechanical Indices for Rupture Risk Estimation in Abdominal Aortic Aneurysms.J Endovasc Ther. 2017 Apr;24(2):254-261. doi: 10.1177/1526602816680088. Epub 2016 Nov 21. J Endovasc Ther. 2017. PMID: 27872318 Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

