A novel technique for heart-thymus en bloc transplantation in nonhuman primates
- PMID: 39738516
- PMCID: PMC11686112
- DOI: 10.1038/s41598-024-83378-4
A novel technique for heart-thymus en bloc transplantation in nonhuman primates
Abstract
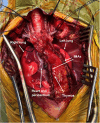
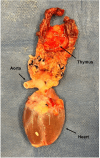
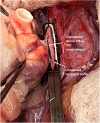
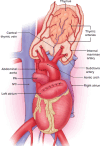
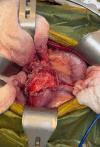
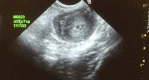
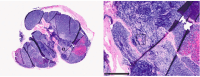
The thymus is a rich source of regulatory T cells and plays a role in self-tolerance. Therefore, transplantation of a vascularized donor thymus may facilitate the induction of tolerance in recipients of a cotransplanted heart allograft. To investigate this hypothesis, we developed a new technique to procure the heart and thymus en bloc from juvenile donors and transplant the composite allograft into thymectomized recipients. Thymic function was monitored by serial biopsy and flow cytometry of peripheral blood. Heart-thymus en bloc transplantation resulted in immediate revascularization of the heart and donor thymus with maintenance of normal thymic architecture, even in biopsies taken months after transplantation. Heart-thymus en bloc transplantation requires minimal modification to current heart procurement techniques. Here, we describe the details of the preparation, procurement, transplantation, and postoperative monitoring for this model, with the intention that this technique could be implemented by other investigators to study the effects of heart and thymus cotransplantation. This method could ultimately offer a new approach to tolerance induction in children.
Keywords: Heart transplant; Pediatric heart transplant; Regulatory T cells; Thymus transplant; Tolerance.
© 2024. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures

References
-
- Chambers, D. C. et al. The international thoracic organ transplant registry of the international society for heart and lung transplantation: thirty-sixth adult lung and heart-lung transplantation report-2019; focus theme: Donor and recipient size match. J. Heart Lung. Transplant.38(10), 1042–1055. 10.1016/j.healun.2019.08.001 (2019). - PMC - PubMed
-
- Kawai, T. et al. CD154 blockade for induction of mixed chimerism and prolonged renal allograft survival in nonhuman primates. Am. J. Transplant.4(9), 1391–1398. 10.1111/j.1600-6143.2004.00523.x (2004). - PubMed
-
- Kawai, T. et al. Mixed allogeneic chimerism and renal allograft tolerance in cynomolgus monkeys. Transplantation.59(2), 256–262 (1995). - PubMed
-
- Kawai, T. et al. Long-term outcome and alloantibody production in a non-myeloablative regimen for induction of renal allograft tolerance. Transplantation.68(11), 1767–1775. 10.1097/00007890-199912150-00022 (1999). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

