Mindin regulates fibroblast subpopulations through distinct Src family kinases during fibrogenesis
- PMID: 39739417
- PMCID: PMC11948575
- DOI: 10.1172/jci.insight.173071
Mindin regulates fibroblast subpopulations through distinct Src family kinases during fibrogenesis
Abstract
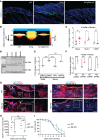
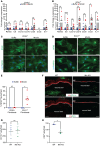
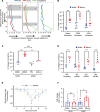
Fibrosis results from excessive extracellular matrix (ECM) deposition, which causes tissue stiffening and organ dysfunction. Activated fibroblasts, central to fibrosis, exhibit increased migration, proliferation, contraction, and ECM production. However, it remains unclear if the same fibroblast performs all of the processes that fall under the umbrella term of "activation." Owing to fibroblast heterogeneity in connective tissues, subpopulations with specific functions may operate under distinct regulatory controls. Using a transgenic mouse model of skin fibrosis, we found that Mindin (also known as spondin-2), secreted by Snail-transgenic keratinocytes, differentially regulates fibroblast subpopulations. Mindin promotes migration and inflammatory gene expression in SCA1+ dermal fibroblasts via Fyn kinase. In contrast, it enhances contractility and collagen production in papillary CD26+ fibroblasts through c-Src signaling. Moreover, in the context of the fibrotic microenvironment of the tumor stroma, we found that differential responses of resident fibroblast subpopulations to Mindin extend to the generation of functionally heterogeneous cancer-associated fibroblasts. This study identifies Mindin as a key orchestrator of dermal fibroblast heterogeneity, reshaping cellular dynamics and signaling diversity in the complex landscapes of skin fibrosis and cancer.
Keywords: Autoimmune diseases; Dermatology; Fibrosis; Inflammation; Signal transduction.
Conflict of interest statement
Figures


Similar articles
-
Mindin (SPON2) Is Essential for Cutaneous Fibrogenesis in a Mouse Model of Systemic Sclerosis.J Invest Dermatol. 2023 May;143(5):699-710.e10. doi: 10.1016/j.jid.2022.10.011. Epub 2022 Dec 15. J Invest Dermatol. 2023. PMID: 36528128
-
Macrophage promotes fibroblast activation and kidney fibrosis by assembling a vitronectin-enriched microenvironment.Theranostics. 2023 Jul 3;13(11):3897-3913. doi: 10.7150/thno.85250. eCollection 2023. Theranostics. 2023. PMID: 37441594 Free PMC article.
-
Sustained β-catenin activity in dermal fibroblasts promotes fibrosis by up-regulating expression of extracellular matrix protein-coding genes.J Pathol. 2015 Apr;235(5):686-97. doi: 10.1002/path.4481. Epub 2015 Jan 7. J Pathol. 2015. PMID: 25385294 Free PMC article.
-
Understanding the impact of fibroblast heterogeneity on skin fibrosis.Dis Model Mech. 2020 Jun 15;13(6):dmm044164. doi: 10.1242/dmm.044164. Dis Model Mech. 2020. PMID: 32541065 Free PMC article. Review.
-
Evidence of fibroblast heterogeneity and the role of fibroblast subpopulations in fibrosis.Clin Immunol Immunopathol. 1994 Sep;72(3):283-92. doi: 10.1006/clin.1994.1144. Clin Immunol Immunopathol. 1994. PMID: 7914840 Review.
References
-
- Thiruvoth FM, et al. Current concepts in the physiology of adult wound healing. Plast Aesthet Res. 2015;2:250–256. doi: 10.4103/2347-9264.158851. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

