Cysteine Leukotriene Receptor Antagonist-Montelukast-Treatment Improves Experimental Abdominal Aortic Aneurysms in Mice
- PMID: 39742015
- PMCID: PMC11617055
- DOI: 10.1155/2024/8826287
Cysteine Leukotriene Receptor Antagonist-Montelukast-Treatment Improves Experimental Abdominal Aortic Aneurysms in Mice
Abstract
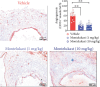
Background: Cysteinyl leukotrienes (LTs) and their receptors are involved in the pathogenesis of abdominal aortic aneurysms (AAAs). However, whether CysLT1 receptor antagonists such as montelukast can influence experimental nondissecting AAA remains unclear. Methods: Nondissecting AAAs were induced in C57BL/6J mice by transient aortic luminal infusion of porcine pancreatic elastase (PPE). All animals were administrated montelukast (1 or 10 mg/kg, daily) or vehicle by gavage beginning 1 day before PPE infusion for 14 days. On day 0 (baseline) and day 14 after PPE infusion, abdominal aortic diameters were directly measured. Aortic aneurysmal segment samples were collected, and histopathological analysis was performed. Results: Compared to vehicle treatment, montelukast significantly decreased PPE infusion-induced aortic expansion in a dose-dependent manner (0.09-mm reduction at a low dose and 0.19-mm reduction at a high dose). Histopathological analysis also revealed that compared with vehicle treatment, montelukast treatment, especially in the high-dose group, significantly improved PPE-induced aortic elastin degradation and medial smooth muscle cell depletion. Both doses of montelukast also markedly decreased the number of local leucocytes, including macrophages, CD4+ T cells, CD8+ T cells, and B cells, infiltration and accumulation in aortic aneurysmal lesions. Montelukast treatment also downregulated matrix metalloproteinase 2 (MMP2) and MMP9 expression and inhibited mural angiogenesis in aneurysmal aortas. Conclusion: Montelukast treatment improves experimental nondissected AAAs in mice partly by improving aortic inflammation.
Keywords: abdominal aortic aneurysm; elastin; inflammation; montelukast; smooth muscle cells.
Copyright © 2024 Kexin Li et al.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




Similar articles
-
Hypoxia-inducible factor 1 in clinical and experimental aortic aneurysm disease.J Vasc Surg. 2018 Nov;68(5):1538-1550.e2. doi: 10.1016/j.jvs.2017.09.030. Epub 2017 Dec 11. J Vasc Surg. 2018. PMID: 29242064 Free PMC article.
-
Dapagliflozin Ameliorates the Formation and Progression of Experimental Abdominal Aortic Aneurysms by Reducing Aortic Inflammation in Mice.Oxid Med Cell Longev. 2022 Jan 28;2022:8502059. doi: 10.1155/2022/8502059. eCollection 2022. Oxid Med Cell Longev. 2022. PMID: 35126822 Free PMC article.
-
Cysteinyl leukotriene receptor 1 antagonism prevents experimental abdominal aortic aneurysm.Proc Natl Acad Sci U S A. 2018 Feb 20;115(8):1907-1912. doi: 10.1073/pnas.1717906115. Epub 2018 Feb 5. Proc Natl Acad Sci U S A. 2018. PMID: 29432192 Free PMC article.
-
Targeting cysteinyl-leukotrienes in abdominal aortic aneurysm.Prostaglandins Other Lipid Mediat. 2018 Nov;139:24-28. doi: 10.1016/j.prostaglandins.2018.09.007. Epub 2018 Sep 21. Prostaglandins Other Lipid Mediat. 2018. PMID: 30248405 Review.
-
Dysregulation of the leukotriene pathway in Parkinson's disease: The potential role of montelukast.Cytokine. 2025 Oct;194:157010. doi: 10.1016/j.cyto.2025.157010. Epub 2025 Aug 13. Cytokine. 2025. PMID: 40812140 Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

