Evaluation of the Efficacy of Lusutrombopag for Chronic Liver Disease Based on Pre-Treatment Platelet Counts: A Retrospective Multicenter Study
- PMID: 39742152
- PMCID: PMC11686089
- DOI: 10.1002/jgh3.70081
Evaluation of the Efficacy of Lusutrombopag for Chronic Liver Disease Based on Pre-Treatment Platelet Counts: A Retrospective Multicenter Study
Abstract
Background: Oral thrombopoietin receptor agonists are used to treat thrombocytopenia in patients with chronic liver disease who are scheduled for invasive procedures. The efficacy of lusutrombopag based on the pretreatment platelet count was investigated.
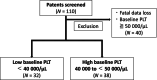
Methods: Patients treated at nine hospitals from December 2015 to December 2023 were included. Efficacy was assessed by comparing the proportion of patients achieving a platelet count ≥ 50 000/μL and the change in platelet count.
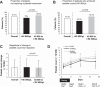
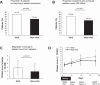
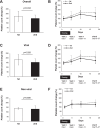
Results: Seventy patients were eligible for evaluation. Patients with a pretreatment platelet count < 40 000/μL had a significantly lower rate of achieving a platelet count of ≥ 50 000/μL than those with a pretreatment count of 40 000-50 000/μL (62.5% vs. 84.2%, p = 0.038); however, there was no significant difference in the change in platelet count (25 700 vs. 24 400/μL, p = 0.972). Patients with viral-related cirrhosis showed a significantly greater change in platelet count than the others (29 100 vs. 19 200/μL, p = 0.012). For patients receiving multiple lusutrombopag treatments, the change in platelet count was significantly lower in the second treatment than in the first treatment (26 900 vs. 20 800/μL, p = 0.041). The main adverse event observed was thrombosis (2.9%).
Discussion: Lusutrombopag increases platelet count regardless of pretreatment levels, but efficacy, defined as achieving a platelet count of ≥ 50 000/μL, may be insufficient in patients with a pretreatment platelet count < 40 000/μL. Additionally, patients with non-viral liver disease responded less well to treatment compared to those with viral liver disease. Therefore, treatment strategies should be tailored based on pretreatment platelet counts and the etiology of liver disease.
Keywords: chronic liver disease; lusutrombopag; thrombocytopenia.
© 2024 The Author(s). JGH Open published by Journal of Gastroenterology and Hepatology Foundation and John Wiley & Sons Australia, Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
-
- Afdhal N., McHutchison J., Brown R., et al., “Thrombocytopenia Associated With Chronic Liver Disease,” Journal of Hepatology 48 (2008): 1000–1007. - PubMed
-
- Dieterich D. T., Bernstein D., Flamm S., Pockros P. J., and Reau N., “Review Article: A Treatment Algorithm for Patients With Chronic Liver Disease and Severe Thrombocytopenia Undergoing Elective Medical Procedures in the United States,” Alimentary Pharmacology & Therapeutics 52 (2020): 1311–1322. - PubMed
LinkOut - more resources
Full Text Sources

