TBK1 inhibitor amlexanox exerts anti-cancer effects against endometrial cancer by regulating AKT/NF-κB signaling
- PMID: 39744441
- PMCID: PMC11667804
- DOI: 10.7150/ijbs.100212
TBK1 inhibitor amlexanox exerts anti-cancer effects against endometrial cancer by regulating AKT/NF-κB signaling
Abstract
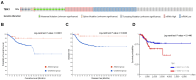
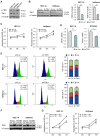
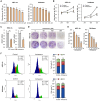
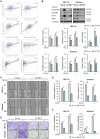
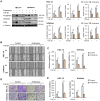
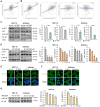
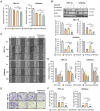
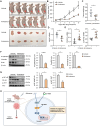
Endometrial cancer, a common gynecological malignancy, poses significant clinical challenges, particularly in advanced or recurrent cases. TANK-binding kinase 1 (TBK1), a serine/threonine kinase, plays crucial roles in inflammation and immunity by activating nuclear factor (NF)-κB and interferon regulatory factor 3. However, its specific roles in endometrial cancer remain unknown. In this study, we aimed to investigate the anti-cancer effects and underlying mechanisms of amlexanox, a TBK1 inhibitor, against endometrial cancer. The main genetic mutations in TBK1 were found to be mRNA downregulation and missense mutations. Kaplan-Meier plotter analysis revealed that low TBK1 expression was associated with a good prognosis in patients with uterine corpus endometrial carcinoma (UCEC). In vitro experiments demonstrated that TBK1 knockdown or amlexanox significantly inhibited the proliferation, cell cycle progression, and migration of endometrial cancer cells. Furthermore, the inhibitory effects of targeting TBK1 on cancer cell proliferation and migration were mediated by the protein kinase B (AKT)/NF-κB signaling pathway. Xenograft experiments revealed that both amlexanox treatment and TBK1 knockdown effectively suppressed the tumor growth. Overall, this study highlights the potent anti-cancer effects of amlexanox against endometrial cancer by modulating AKT/NF-κB signaling, thus providing a new avenue for the development of novel TBK1-targeting therapeutic strategies for UCEC.
Keywords: NF-κB; TBK1; endometrial cancer; prognosis; shRNA; xenograft.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures
References
-
- Crosbie EJ, Kitson SJ, McAlpine JN, Mukhopadhyay A, Powell ME, Singh N. Endometrial cancer. Lancet. 2022;399:1412–28. - PubMed
-
- Group SGOCPECW, Burke WM, Orr J, Leitao M, Salom E, Gehrig P. et al. Endometrial cancer: a review and current management strategies: part I. Gynecol Oncol. 2014;134:385–92. - PubMed
-
- Bouwman F, Smits A, Lopes A, Das N, Pollard A, Massuger L. et al. The impact of BMI on surgical complications and outcomes in endometrial cancer surgery-an institutional study and systematic review of the literature. Gynecol Oncol. 2015;139:369–76. - PubMed
-
- Helgason E, Phung QT, Dueber EC. Recent insights into the complexity of Tank-binding kinase 1 signaling networks: the emerging role of cellular localization in the activation and substrate specificity of TBK1. FEBS Lett. 2013;587:1230–7. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

