The D-lactate enigma: exploring the inflammatory influence of D-lactate in cattle
- PMID: 39744722
- PMCID: PMC11688252
- DOI: 10.3389/fvets.2024.1509399
The D-lactate enigma: exploring the inflammatory influence of D-lactate in cattle
Abstract
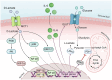
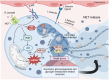
D-lactic acidosis is associated with fermentative disturbances and is often marked by elevated levels of D-lactic acid in the blood, ruminal fluid, and synovial fluid in cattle. D-lactic acidosis is linked to various inflammatory manifestations, and although the causative factors have been extensively explored, the exact pathogenesis of the associated inflammation remains elusive. Notably, less attention has been given to D-lactate, a stereoisomer found in the plasma of affected animals, which may lead to D-lactic acidosis. This review aims to highlight the evidence suggesting that D-lactate participates in the modulation of inflammatory processes and explore its potential effects on synoviocytes, polymorphonuclear neutrophils, macrophages, and T-cells. This comprehensive examination of D-lactate's involvement in the inflammatory response process provides timely insights into the pathophysiological aspects of ruminal acidosis in cattle.
Keywords: D-lactate; bovine; inflammation; lameness; ruminal acidosis.
Copyright © 2024 Burgos, Manosalva, Alarcón, Navarro, Quiroga, Morán, Gallastegui, Brauchi and Carretta.
Conflict of interest statement
The authors declare that this review was conducted without any commercial or financial relationships that could be construed as potential conflicts of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

