Deciphering the role of APOE in cerebral amyloid angiopathy: from genetic insights to therapeutic horizons
- PMID: 39745195
- PMCID: PMC11703089
- DOI: 10.1080/07853890.2024.2445194
Deciphering the role of APOE in cerebral amyloid angiopathy: from genetic insights to therapeutic horizons
Abstract
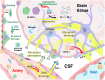
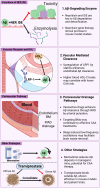
Cerebral amyloid angiopathy (CAA), characterized by the deposition of amyloid-β (Aβ) peptides in the walls of medium and small vessels of the brain and leptomeninges, is a major cause of lobar hemorrhage in elderly individuals. Among the genetic risk factors for CAA that continue to be recognized, the apolipoprotein E (APOE) gene is the most significant and prevalent, as its variants have been implicated in more than half of all patients with CAA. While the presence of the APOE ε4 allele markedly increases the risk of CAA, the ε2 allele confers a protective effect relative to the common ε3 allele. These allelic variants encode three APOE isoforms that differ at two amino acid positions. The primary physiological role of APOE is to mediate lipid transport in the brain and periphery; however, it has also been shown to be involved in a wide array of biological functions, particularly those involving Aβ, in which it plays a known role in processing, production, aggregation, and clearance. The challenges posed by the reliance on postmortem histological analyses and the current absence of an effective intervention underscore the urgency for innovative APOE-targeted strategies for diagnosing CAA. This review not only deepens our understanding of the impact of APOE on the pathogenesis of CAA but can also help guide the exploration of targeted therapies, inspiring further research into the therapeutic potential of APOE.
Keywords: Cerebral amyloid angiopathy; amyloid β; apolipoprotein E; diagnosis; pathology; therapy.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures
Similar articles
-
Apolipoprotein E-epsilon4 alleles in cerebral amyloid angiopathy and cerebrovascular pathology associated with Alzheimer's disease.Am J Pathol. 1996 Jun;148(6):2083-95. Am J Pathol. 1996. PMID: 8669492 Free PMC article.
-
Two types of sporadic cerebral amyloid angiopathy.J Neuropathol Exp Neurol. 2002 Mar;61(3):282-93. doi: 10.1093/jnen/61.3.282. J Neuropathol Exp Neurol. 2002. PMID: 11895043
-
Human apolipoprotein E4 alters the amyloid-beta 40:42 ratio and promotes the formation of cerebral amyloid angiopathy in an amyloid precursor protein transgenic model.J Neurosci. 2005 Mar 16;25(11):2803-10. doi: 10.1523/JNEUROSCI.5170-04.2005. J Neurosci. 2005. PMID: 15772340 Free PMC article.
-
Role of apoe/Abeta interactions in the pathogenesis of Alzheimer's disease and cerebral amyloid angiopathy.J Mol Neurosci. 2001 Oct;17(2):147-55. doi: 10.1385/JMN:17:2:147. J Mol Neurosci. 2001. PMID: 11816788 Review.
-
Apolipoprotein E genotype and cerebral amyloid angiopathy-related hemorrhage.Ann N Y Acad Sci. 2000 Apr;903:176-9. doi: 10.1111/j.1749-6632.2000.tb06366.x. Ann N Y Acad Sci. 2000. PMID: 10818505 Review.
Cited by
-
CHIT1 and DDAH1 levels relate to amyloid-related imaging abnormalities risk profile in Alzheimer's disease patients.Alzheimers Res Ther. 2025 Jul 22;17(1):169. doi: 10.1186/s13195-025-01799-3. Alzheimers Res Ther. 2025. PMID: 40696469 Free PMC article.
-
Clinical Management of Cerebral Amyloid Angiopathy.J Clin Med. 2025 Jun 15;14(12):4259. doi: 10.3390/jcm14124259. J Clin Med. 2025. PMID: 40566003 Free PMC article. Review.
-
APOE Genotyping in Cognitive Disorders: Preliminary Observations from the Greek Population.Int J Mol Sci. 2025 Aug 1;26(15):7410. doi: 10.3390/ijms26157410. Int J Mol Sci. 2025. PMID: 40806539 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
