Genetic identification of acetyl-CoA synthetases involved in acetate activation in Haloferax mediterranei
- PMID: 39745370
- PMCID: PMC11784441
- DOI: 10.1128/aem.01843-24
Genetic identification of acetyl-CoA synthetases involved in acetate activation in Haloferax mediterranei
Abstract
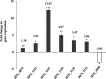
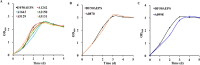
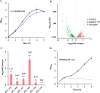
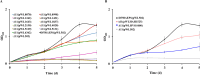
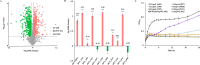
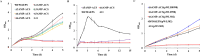
Acetate/acetyl-CoA interconversion is an interesting metabolic node, primarily catalyzed by a set of various enzymes in prokaryotes. Haloferax mediterranei is a promising haloarchaeaon, capable of utilizing acetate as a sole carbon source for biosynthesis of high value-added products. Here, we have reported the key enzymes that catalyzed acetate activation in H. mediterranei. Based on bioinformatic and transcript analysis, thirteen possible candidate genes were screened. Simultaneous deletion of eleven genes led to a mutant strain (named as Δ11) that failed to grow on acetate. Gene complementation in Δ11 revealed six AMP-ACS (encoded by HFX_0870, HFX_1242, HFX_1451, HFX_6342, HFX_5131, and HFX_1643) and one ADP-ACS (encoded by HFX_0998) to be functional in acetate activation. Furthermore, heterologous expression of ADP-ACS genes from Haloarcula hispanica and Haloferax volcanii catalyzed acetate activation in Δ11. Subsequently, it was observed that, deletion of the six AMP-ACS genes in H. mediterranei ceased the cell growth of the resulting mutant (Δ6AMP-ACS) on acetate. An in vivo function of ADP-ACS in acetate activation could be excluded since ADP-ACS was downregulated on acetate. However, plasmid-based overexpression of ADP-ACS enabled Δ6AMP-ACS to grow on acetate, even better than the parent strain. Thus, it can be inferred that native ADP-ACS with low expression level was unable to mediate cell growth of Δ6AMP-ACS on acetate. This is the first genetic evidence exhibiting that overexpression of haloarchaeal ADP-ACS catalyzed acetate activation in vivo. Collectively, this is a comprehensive study of acetate activation in H. mediterranei, and the current findings would surely enrich the understanding of acetate metabolism in archaea.
Importance: Owing to the high demand and supply challenge of glucose, acetate might be considered a potential alternative carbon source for microbial growth and fermentation. Haloferax mediterranei is capable of utilizing acetate as a carbon source for growth and subsequent value-added product synthesis. Thus, it is essential to identify the genes responsible for acetate utilization in H. mediterranei. As per available literature, haloarchaeal ADP-forming acetyl-CoA synthetase (APD-ACS) catalyzes the reversible conversion of acetate to acetyl-CoA in vitro. However, in vivo, acetate activation and acetate formation are catalyzed by AMP-forming acetyl-CoA synthetase (AMP-ACS) and ADP-ACS, respectively. In this study, we have identified six AMP-ACS enzymes that catalyzed acetate activation in H. mediterranei. Deletion of these six genes abolished the growth of the resulting mutant (Δ6AMP-ACS) in acetate medium. The natively expressed ADP-ACS was unable to mediate its acetate activation in vivo. Interestingly, an artificial system based on plasmid overexpression of ADP-ACS in Δ6AMP-ACS restored its growth on acetate. This finding suggested that native ADP-ACS was unable to catalyze acetate activation in H. mediterranei due to its low expression level. Together, our study explored the acetate activation in H. mediterranei, and the obtained results would enrich the knowledge of acetate metabolism in archaea. Furthermore, the information offered in this study would benefit the improvement of acetate utilization in haloarchaea for value-added product synthesis.
Keywords: ADP-acetyl-CoA synthetase; AMP-acetyl-CoA synthetase; acetate activation; haloarchaea.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Qin C, Graf LG, Striska K, Janetzky M, Geist N, Specht R, Schulze S, Palm GJ, Girbardt B, Dörre B, Berndt L, Kemnitz S, Doerr M, Bornscheuer UT, Delcea M, Lammers M. 2024. Acetyl-CoA synthetase activity is enzymatically regulated by lysine acetylation using acetyl-CoA or acetyl-phosphate as donor molecule. Nat Commun 15:6002. doi: 10.1038/s41467-024-49952-0 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

