Microfluidic 3D cell culture: potential application of collagen hydrogels with an optimal dose of bioactive glasses
- PMID: 39747624
- PMCID: PMC11696724
- DOI: 10.1038/s41598-024-84346-8
Microfluidic 3D cell culture: potential application of collagen hydrogels with an optimal dose of bioactive glasses
Abstract
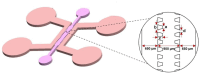
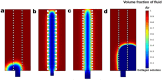
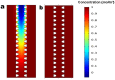
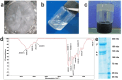
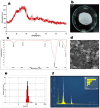
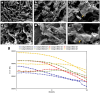
We engineered a microfluidic platform to study the effects of bioactive glass nanoparticles (BGNs) on cell viability under static culture. We incorporated different concentrations of BGNs (1%, 2%, and 3% w/v) in collagen hydrogel (with a concentration of 3.0 mg/mL). The microfluidic chip's dimensions were optimized through fluid flow and mass transfer simulations. Collagen type I extracted from rat tail tendons was used as the main material, and BGNs synthesized by the sol-gel method were used to enhance the mechanical properties of the hydrogel. The extracted collagen was characterized using FTIR and SDS-PAGE, and BGNs were analyzed using XRD, FTIR, DLS, and FE-SEM/EDX. The structure of the collagen-BGNs hydrogels was examined using SEM, and their mechanical properties were determined using rheological analysis. The cytotoxicity of BGNs was assessed using the MTT assay, and the viability of fibroblast (L929) cells encapsulated in the collagen-BGNs hydrogel inside the microfluidic device was assessed using a live/dead assay. Based on all these test results, the L929 cells showed high cell viability in vitro and promising microenvironment mimicry in a microfluidic device. Collagen3-BGNs3 (Collagen 3 mg/mL + BGNs 3% (w/v)) was chosen as the most suitable sample for further research on a microfluidic platform.
Keywords: 3D cell culture; Bioactive glass nanoparticle; Collagen hydrogel; Microfluidic system.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests. Ethical approval: The research ethics committee of Iran University of Medical Sciences, Tehran, Iran approved this investigation (Ethical Code: IR.IUMS.REC.1402.489). All experiments were performed according to relevant guidelines and regulations approved by the Research Ethics Committee of Iran University Medical of Sciences, Tehran, Iran. The animal experiment related to collagen extraction from rat tail tendons followed the ARRIVE guidelines.
Figures




Similar articles
-
Mg-Sr-Ca containing bioactive glass nanoparticles hydrogel modified mineralized collagen scaffold for bone repair.J Biomater Appl. 2024 Aug;39(2):117-128. doi: 10.1177/08853282241254741. Epub 2024 May 22. J Biomater Appl. 2024. PMID: 38775351
-
Thermogelling chitosan-collagen-bioactive glass nanoparticle hybrids as potential injectable systems for tissue engineering.Mater Sci Eng C Mater Biol Appl. 2016 Jan 1;58:1207-16. doi: 10.1016/j.msec.2015.09.075. Epub 2015 Sep 25. Mater Sci Eng C Mater Biol Appl. 2016. PMID: 26478423
-
Photo-crosslinkable hydrogel-based 3D microfluidic culture device.Electrophoresis. 2015 Apr;36(7-8):994-1001. doi: 10.1002/elps.201400465. Epub 2015 Mar 24. Electrophoresis. 2015. PMID: 25641332
-
Engineered cell-laden alginate microparticles for 3D culture.Biochem Soc Trans. 2021 Apr 30;49(2):761-773. doi: 10.1042/BST20200673. Biochem Soc Trans. 2021. PMID: 33860783 Free PMC article. Review.
-
Microfluidic technologies for vasculature biomimicry.Analyst. 2019 Jul 22;144(15):4461-4471. doi: 10.1039/c9an00421a. Analyst. 2019. PMID: 31162494 Review.
References
-
- Bhatt, A. et al. Biocompatibility-on-a-chip: characterization and evaluation of decellularized tendon extracellular matrix (tdECM) hydrogel for 3D stem cell culture in a microfluidic device. Int. J. Biol. Macromol.213, 768–779. 10.1016/J.IJBIOMAC.2022.06.010 (2022). - PubMed
-
- Lee, P., Lin, R., Moon, J. & Lee, L. P. Microfluidic alignment of collagen fibers for in vitro cell culture. Biomed. Microdevices.8(1), 35–41. 10.1007/S10544-006-6380-Z/METRICS (2006). - PubMed
-
- Chen, G., Sato, T., Ushida, T., Ochiai, N. & Tateishi, T. Tissue engineering of cartilage using a hybrid scaffold of synthetic polymer and collagen. Tissue Eng.10(3–4), 323–330. 10.1089/107632704323061681 (2004). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

