On the Role of Hydrogen Migrations in the Taxadiene System
- PMID: 39749413
- PMCID: PMC11933516
- DOI: 10.1002/anie.202422788
On the Role of Hydrogen Migrations in the Taxadiene System
Abstract
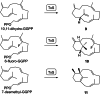
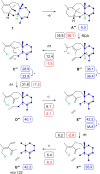
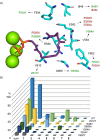
Taxa-4,11-diene is made by the taxa-4,11-diene synthase (TxS) from Taxus brevifolia. The unique reactivity of the taxane system is characterised by long distance hydrogen migrations in the biosynthesis. This study demonstrates that selective long range hydrogen migrations also play a role in the high energy process of the EI-MS fragmentation of taxa-4,11-diene. A TxS enzyme variant was generated that produces cyclophomactene, a compound that is formed through a concerted process involving a long range proton shift and a ring closure that can also be described as the addition of a methylcarbinyl cation to an olefin. Based on a previous computational study the cyclisation mechanism towards taxa-4,11-diene was suggested to involve two long distance proton migrations instead of one direct transfer. A substrate analog with a shifted double bond was converted with TxS to obtain experimental evidence for this proposal.
Keywords: biosynthesis; enzymes; natural products; substrate analogs; terpenoids.
© 2025 The Author(s). Angewandte Chemie International Edition published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Intramolecular proton transfer in the cyclization of geranylgeranyl diphosphate to the taxadiene precursor of taxol catalyzed by recombinant taxadiene synthase.Chem Biol. 2000 Dec;7(12):969-77. doi: 10.1016/s1074-5521(00)00046-6. Chem Biol. 2000. PMID: 11137819
-
Mechanism of taxadiene synthase, a diterpene cyclase that catalyzes the first step of taxol biosynthesis in Pacific yew.Biochemistry. 1996 Mar 5;35(9):2968-77. doi: 10.1021/bi9526239. Biochemistry. 1996. PMID: 8608134
-
Taxadiene synthase structure and evolution of modular architecture in terpene biosynthesis.Nature. 2011 Jan 6;469(7328):116-20. doi: 10.1038/nature09628. Epub 2010 Dec 15. Nature. 2011. PMID: 21160477 Free PMC article.
-
The verticillenes. Pivotal intermediates in the biosynthesis of the taxanes and the phomactins.Nat Prod Rep. 2019 Jan 1;36(1):108-121. doi: 10.1039/c8np00034d. Epub 2018 Jul 6. Nat Prod Rep. 2019. PMID: 29978865 Review.
-
Manganese-Oxygen Intermediates in O-O Bond Activation and Hydrogen-Atom Transfer Reactions.Acc Chem Res. 2017 Nov 21;50(11):2706-2717. doi: 10.1021/acs.accounts.7b00343. Epub 2017 Oct 24. Acc Chem Res. 2017. PMID: 29064667 Review.
Cited by
-
Stereoselective deprotonation installs an unusual Z-8,9 double bond during biosynthesis of the diterpene pheromone sobralene.Chem Commun (Camb). 2025 Aug 5;61(64):12026-12029. doi: 10.1039/d5cc03298a. Chem Commun (Camb). 2025. PMID: 40641443 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

