TRIOL attenuates intracerebral hemorrhage injury by bidirectionally modulating microglia- and neuron-mediated hematoma clearance
- PMID: 39756315
- PMCID: PMC11758845
- DOI: 10.1016/j.redox.2024.103487
TRIOL attenuates intracerebral hemorrhage injury by bidirectionally modulating microglia- and neuron-mediated hematoma clearance
Abstract
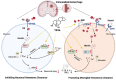
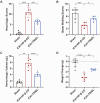
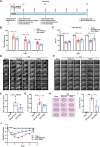
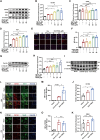
Intracerebral hemorrhage (ICH) represents the most severe subtype of stroke, and the lack of effective clinical pharmacotherapies poses a substantial threat to human health. Hematoma plays a crucial role in determining the prognosis of ICH patients by causing primary mechanical extrusion, followed by secondary brain injuries, such as cerebral edema, iron-mediated oxidative stress, and inflammation resulting from its degradation products. 5α-androst-3β,5α,6β-triol (TRIOL) is a neuroprotective steroid currently undergoing phase II clinical trial for acute ischemic stroke with anti-oxidative and anti-inflammatory properties. However, whether TRIOL can protect brain against ICH injury remains unclear. In this study, we found that TRIOL significantly improved neurological function while reducing hematoma volume, cerebral edema, and tissue damage after ICH. Moreover, TRIOL enhanced microglial hematoma clearance through promoting CD36-mediated erythrophagocytosis and CD163-associated hemoglobin scavenging, while simultaneously reducing the release of microglial inflammatory factors and activating the antioxidative transcription factor Nrf2. Additionally, TRIOL inhibited neuron mediated hematoma absorption by suppressing heme oxygenase 2 (HO-2) and protected neurons against ICH-induced damage in vitro and in vivo. TRIOL also mitigated neuronal iron-dependent oxidative damage by increasing ferritin levels but decreasing divalent metal transporter 1 (DMT1) expression. Overall, these findings highlight the promising potential of TRIOL as a drug candidate for treating ICH.
Keywords: Hematoma clearance; Heme oxygenase 1; Heme oxygenase 2; Intracerebral hemorrhage; Nrf2; TRIOL.
Copyright © 2024 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare no competing interest.
Figures





References
-
- Puy L., Parry-Jones A.R., Sandset E.C., Dowlatshahi D., Ziai W., Cordonnier C. Intracerebral haemorrhage. Nat. Rev. Dis. Prim. 2023;9(1):14. - PubMed
-
- Broderick J.P., Brott T.G., Duldner J.E., Tomsick T., Huster G. Volume of intracerebral hemorrhage. A powerful and easy-to-use predictor of 30-day mortality. Stroke. 1993;24(7):987–993. - PubMed
-
- Greenberg S.M., Ziai W.C., Cordonnier C., Dowlatshahi D., Francis B., Goldstein J.N., et al. Guideline for the management of patients with spontaneous intracerebral hemorrhage: a guideline from the American heart association/American stroke association. Stroke. 2022;53(7):e282–e361. 2022. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

