Diversification of Lipopeptide Analogues Drives Versatility in Biological Activities
- PMID: 39760433
- PMCID: PMC11741111
- DOI: 10.1021/acs.jafc.4c11372
Diversification of Lipopeptide Analogues Drives Versatility in Biological Activities
Abstract
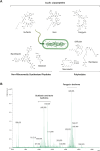
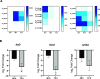
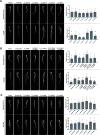
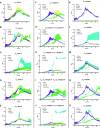
Cyclic lipopeptides (CLPs) are potent secondary metabolites with diverse biological functions. Bacillus strains primarily produce CLPs of three key families, namely, iturins, fengycins, and surfactins, each comprising structural variants characterized by a cyclic peptide linked to a fatty acid chain. Despite extensive research on CLPs, the individual roles of these analogues and their proportion in driving biological activity have remained largely overlooked. In this study, we purified and chemically characterized CLP variants from Bacillus velezensis UMAF6639 and tested them individually for their antifungal and plant growth-promoting effects. We isolated 5 fractions containing iturin A analogues (from C13 to C17), 5 fengycin fractions (containing C16, C17, and C18 fengycin A and C14, C15, C16, and C17 fengycin B), and 5 surfactin fractions (from C12 to C16). We show how antifungal activity and seed radicle growth promotion relied on the lipopeptide structural variant and concentration based on the physiological ratio calculated for each lipopeptide variant. Notably, we found that the most toxic variants were the least abundant, which likely minimized autotoxicity while preserving bioactivity. This balance is achieved through synergistic interactions with more abundant, less aggressive analogues. Furthermore, certain fengycin and surfactin variants were shown to increase bacterial population density and exopolysaccharide production, crucial strategies for microbial competition with significant ecological impacts. In addition to advancing basic knowledge, our findings will support the development of precision biotechnological innovations, offering targeted solutions to drive sustainable food production and preservation strategies.
Keywords: Bacillus velezensis; analogues; antifungal; biotechnology; cyclic lipopeptides; food control.; plant growth promotion; structural variants; sustainable agriculture.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



References
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Miscellaneous

