Tailoring rhodium-based metal-organic layers for parahydrogen-induced polarization: achieving 20% polarization of 1H in liquid phase
- PMID: 39764503
- PMCID: PMC11702662
- DOI: 10.1093/nsr/nwae406
Tailoring rhodium-based metal-organic layers for parahydrogen-induced polarization: achieving 20% polarization of 1H in liquid phase
Abstract
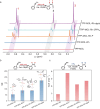
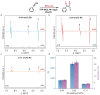
Heterogeneous catalysts for parahydrogen-induced polarization (HET-PHIP) would be useful for producing highly sensitive contrasting agents for magnetic resonance imaging (MRI) in the liquid phase, as they can be removed by simple filtration. Although homogeneous hydrogenation catalysts are highly efficient for PHIP, their sensitivity decreases when anchored on porous supports due to slow substrate diffusion to the active sites and rapid depolarization within the channels. To address this challenge, we explored 2D metal-organic layers (MOLs) as supports for active Rh complexes with diverse phosphine ligands and tunable hydrogenation activities, taking advantage of the accessible active sites and chemical adaptability of the MOLs. By adjusting the electronic properties of phosphines, TPP-MOL-Rh-dppb (TPP = tris(4-carboxylphenyl)phosphine), featuring a κ 2-connected di(phosphine) ligand, generated hyperpolarized styrene achieving an over-2400-fold signal enhancement and a polarization level of 20% for 1H in methanol-d 4 solution. The TPP-MOL-Rh-dppb effectively inherited the high efficiency and pairwise addition of its homogenous catalyst while maintaining the heterogeneity of MOLs. This work demonstrates the potential of 2D phosphine-functionalized MOLs as heterogeneous solid support for HET-PHIP.
Keywords: heterogeneous catalysts; hyperpolarization; metal-organic layers; nuclear magnetic resonance; parahydrogen-induced polarization.
© The Author(s) 2024. Published by Oxford University Press on behalf of China Science Publishing & Media Ltd.
Figures




References
-
- Wang W, Wang Q, Xu J et al. Understanding heterogeneous catalytic hydrogenation by parahydrogen-induced polarization NMR spectroscopy. ACS Catal 2023; 13: 3501–19. 10.1021/acscatal.2c05659 - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous
