Enzymatic Synthesis of Biologically Active H-Phosphinic Analogue of α-Ketoglutarate
- PMID: 39766281
- PMCID: PMC11673680
- DOI: 10.3390/biom14121574
Enzymatic Synthesis of Biologically Active H-Phosphinic Analogue of α-Ketoglutarate
Abstract
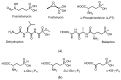
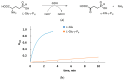
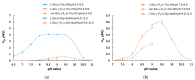
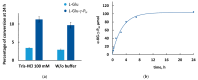
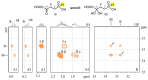
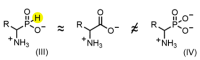
Amino acid analogues with a phosphorus-containing moiety replacing the carboxylic group are promising sources of biologically active compounds. The H-phosphinic group, with hydrogen-phosphorus-carbon (H-P-C) bonds and a flattened tetrahedral configuration, is a bioisostere of the carboxylic group. Consequently, amino-H-phosphinic acids undergo substrate-like enzymatic transformations, leading to new biologically active metabolites. Previous studies employing NMR-based metabolomic and proteomic analyses show that in Escherichia coli, α-KG-γ-PH (the distal H-phosphinic analogue of α-ketoglutarate) can be converted into L-Glu-γ-PH. Notably, α-KG-γ-PH and L-Glu-γ-PH are antibacterial compounds, but their intracellular targets only partially overlap. L-Glu-γ-PH is known to be a substrate of aspartate transaminase and glutamate decarboxylase, but its substrate properties with NAD+-dependent glutamate dehydrogenase (GDH) have never been investigated. Compounds containing P-H bonds are strong reducing agents; therefore, enzymatic NAD+-dependent oxidation is not self-evident. Herein, we demonstrate that L-Glu-γ-PH is a substrate of eukaryotic GDH and that the pH optimum of L-Glu-γ-PH NAD+-dependent oxidative deamination is shifted to a slightly alkaline pH range compared to L-glutamate. By 31P NMR, we observe that α-KG-γ-PH exists in a pH-dependent equilibrium of keto and germinal diol forms. Furthermore, the stereospecific enzymatic synthesis of α-KG-γ-PH from L-Glu-γ-PH using GDH is a possible route for its bio-based synthesis.
Keywords: H-phosphinic analogue of α-ketoglutarate; H-phosphinic analogues of glutamate; glutamate dehydrogenase; glutamate metabolism.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Integrated multi-omics unveil the impact of H-phosphinic analogs of glutamate and α-ketoglutarate on Escherichia coli metabolism.J Biol Chem. 2024 Oct;300(10):107803. doi: 10.1016/j.jbc.2024.107803. Epub 2024 Sep 21. J Biol Chem. 2024. PMID: 39307306 Free PMC article.
-
Enzymatic kinetic resolution of desmethylphosphinothricin indicates that phosphinic group is a bioisostere of carboxyl group.Commun Chem. 2020 Sep 2;3(1):121. doi: 10.1038/s42004-020-00368-z. Commun Chem. 2020. PMID: 36703359 Free PMC article.
-
A Desmethylphosphinothricin Dipeptide Derivative Effectively Inhibits Escherichia coli and Bacillus subtilis Growth.Biomolecules. 2023 Sep 26;13(10):1451. doi: 10.3390/biom13101451. Biomolecules. 2023. PMID: 37892133 Free PMC article.
-
Structural features of aluminium(III) complexes with bioligands in glutamate dehydrogenase reaction system--a review.J Inorg Biochem. 2007 Sep;101(9):1242-50. doi: 10.1016/j.jinorgbio.2007.06.030. Epub 2007 Jun 30. J Inorg Biochem. 2007. PMID: 17643493 Review.
-
Stereoselective Synthesis of α-Amino-C-phosphinic Acids and Derivatives.Molecules. 2016 Aug 29;21(9):1141. doi: 10.3390/molecules21091141. Molecules. 2016. PMID: 27589703 Free PMC article. Review.
References
-
- Pines G., Oh E.J., Bassalo M.C., Choudhury A., Garst A.D., Fankhauser R.G., Eckert C.A., Gill R.T. Genomic deoxyxylulose phosphate reductoisomerase (DXR) mutations conferring resistance to the antimalarial drug fosmidomycin in E. coli. ACS Synth. Biol. 2018;7:2824–2832. doi: 10.1021/acssynbio.8b00219. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

