Bioactive Molecules from the Exoskeleton of Procambarus clarkii: Reducing Capacity, Radical Scavenger, and Antitumor and Anti-Inflammatory Activities
- PMID: 39766342
- PMCID: PMC11726989
- DOI: 10.3390/biom14121635
Bioactive Molecules from the Exoskeleton of Procambarus clarkii: Reducing Capacity, Radical Scavenger, and Antitumor and Anti-Inflammatory Activities
Abstract
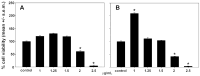
This study evaluates, for the first time, the reducing capacity, radical scavenger activity, and in vitro antitumor and anti-inflammatory effects of chitosan, astaxanthin, and bio-phenols extracted from the exoskeleton of Sicilian Procambarus clarkii, the most widespread species of invasive crayfish in the Mediterranean region. Among the extracted compounds, astaxanthin exhibited the highest antioxidant activity in all assays. Chitosan and polyphenols demonstrated reducing and radical scavenging activity; chitosan showed significant ferric ion reducing capacity in the FRAP test, while bio-phenolic compounds displayed notable radical scavenging activity in the DPPH and ABTS assays. Both astaxanthin and polyphenols showed dose-dependent cytotoxicity on two different cancer cell lines, with IC50 values of 1.45 µg/mL (phenolic extract) and 4.28 µg/mL (astaxanthin extract) for HepG2 cells and 2.45 µg/mL (phenolic extract) and 4.57 µg/mL (astaxanthin extract) for CaCo-2 cells. The bio-phenolic extract also showed potential anti-inflammatory effects in vitro by inhibiting nitric oxide production in inflamed RAW 264.7 macrophages, reducing the treated/control NO ratio to 77% and 74% at concentrations of 1.25 and 1.5 μg/mL, respectively. These results suggest that P. clarkii exoskeletons could be a valuable source of bioactive molecules for biomedical, pharmaceutical, and nutraceutical application while contributing to the sustainable management of this invasive species.
Keywords: antioxidant; astaxanthin; chitosan; crustacean; freshwater; invertebrates; phenolic compounds.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures





References
-
- Ngasotter S., Xavier K.A.M., Meitei M.M., Waikhom D., Madhulika, Pathak J., Singh S.K. Crustacean Shell Waste Derived Chitin and Chitin Nanomaterials for Application in Agriculture, Food, and Health—A Review. Carbohydr. Polym. Technol. Appl. 2023;6:100349. doi: 10.1016/j.carpta.2023.100349. - DOI
-
- Taher F.A., Ibrahim S.A., El-Aziz A.A., Abou El-Nour M.F., El-Sheikh M.A., El-Husseiny N., Mohamed M.M. Anti-Proliferative Effect of Chitosan Nanoparticles (Extracted from Crayfish Procambarus clarkii, Crustacea: Cambaridae) against MDA-MB-231 and SK-BR-3 Human Breast Cancer Cell Lines. Int. J. Biol. Macromol. 2019;126:478–487. doi: 10.1016/j.ijbiomac.2018.12.151. - DOI - PubMed
-
- Ibrahim A.M., Morad M.Y., Hamdi S.A.H., Fol M.F. Biocontrol Potential of Chitosan Extracted from Procambarus clarkii (Crustacea: Cambaridae) against Eobania vermiculata Snails (Muller 1774) in Egypt. Egypt. J. Biol. Pest Control. 2022;32:32. doi: 10.1186/s41938-022-00521-x. - DOI
-
- El-Naggar M.M., Abou-Elmagd W.S.I., Suloma A., El-Shabaka H.A., Khalil M.T., Abd El-Rahman F.A. Optimization and Physicochemical Characterization of Chitosan and Chitosan Nanoparticles Extracted from the Crayfish Procambarus clarkii Wastes. J. Shellfish Res. 2019;38:385. doi: 10.2983/035.038.0220. - DOI
-
- Synowiecki J., Al-Khateeb N.A.A.Q. The Recovery of Protein Hydrolysate during Enzymatic Isolation of Chitin from Shrimp Crangon crangon Processing Discards. Food Chem. 2000;68:147–152. doi: 10.1016/S0308-8146(99)00165-X. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

