iPSC-Derived Biological Pacemaker-From Bench to Bedside
- PMID: 39768137
- PMCID: PMC11674228
- DOI: 10.3390/cells13242045
iPSC-Derived Biological Pacemaker-From Bench to Bedside
Abstract
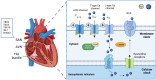
Induced pluripotent stem cell (iPSC)-derived biological pacemakers have emerged as an alternative to traditional electronic pacemakers for managing cardiac arrhythmias. While effective, electronic pacemakers face challenges such as device failure, lead complications, and surgical risks, particularly in children. iPSC-derived pacemakers offer a promising solution by mimicking the sinoatrial node's natural pacemaking function, providing a more physiological approach to rhythm control. These cells can differentiate into cardiomyocytes capable of autonomous electrical activity, integrating into heart tissue. However, challenges such as achieving cellular maturity, long-term functionality, and immune response remain significant barriers to clinical translation. Future research should focus on refining gene-editing techniques, optimizing differentiation, and developing scalable production processes to enhance the safety and effectiveness of these biological pacemakers. With further advancements, iPSC-derived pacemakers could offer a patient-specific, durable alternative for cardiac rhythm management. This review discusses key advancements in differentiation protocols and preclinical studies, demonstrating their potential in treating dysrhythmias.
Keywords: HCN channels; induced pluripotent stem cell; sinoatrial node.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Glikson M., Nielsen J.C., Kronborg M.B., Michowitz Y., Auricchio A., Barbash I.M., Barrabés J.A., Boriani G., Braunschweig F., Brignole M., et al. 2021 ESC Guidelines on cardiac pacing and cardiac resynchronization therapy. Eur. Heart J. 2021;42:3427–3520. doi: 10.1093/eurheartj/ehab364. - DOI - PubMed
-
- Virani S.S., Alonso A., Aparicio H.J., Benjamin E.J., Bittencourt M.S., Callaway C.W., Carson A.P., Chamberlain A.M., Cheng S., Delling F.N., et al. Heart Disease and Stroke Statistics-2021 Update: A Report from the American Heart Association. Circulation. 2021;143:e254–e743. doi: 10.1161/cir.0000000000000950. - DOI - PubMed
-
- de Heide J., van der Graaf M., Holl M.J., Hoogendijk M.G., Bhagwandien R.E., Wijchers S.A., Theuns D., Szili-Torok T., Zijlstra F., Lenzen M.J., et al. Device infection in patients undergoing pacemaker or defibrillator surgery: Risk stratification using the PADIT score. J. Interv. Card. Electrophysiol. 2024;67:1419–1426. doi: 10.1007/s10840-024-01759-1. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

