Assessing Cytotoxicity, Proteolytic Stability, and Selectivity of Antimicrobial Peptides: Implications for Orthopedic Applications
- PMID: 39769006
- PMCID: PMC11678430
- DOI: 10.3390/ijms252413241
Assessing Cytotoxicity, Proteolytic Stability, and Selectivity of Antimicrobial Peptides: Implications for Orthopedic Applications
Abstract
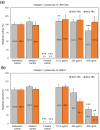
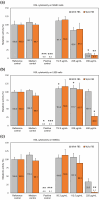
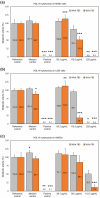
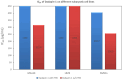
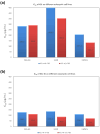
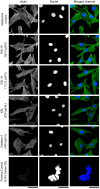
In orthopedics, the use of anti-infective biomaterials is considered the most promising strategy to contrast the bacterial contamination of implant surfaces and reduce the infection rate. KSL, KSL-W, and Dadapin-1 are three antimicrobial peptides (AMPs) that possess significant antibacterial properties, making them promising candidates for producing anti-infective biomaterials not based on antibiotics. To fully assess their true potential, this study explores in detail their cytocompatibility on human osteoblast-like MG63 cells, murine fibroblastoid L929 cells, and hMSCs. To this end, the cytotoxicity of the AMPs in terms of IC50 was tested over a range of concentrations of 450-0.22 µg/mL using the ATP bioluminescence assay. The tests were performed both in the presence and absence of bovine serum to assess the effects of serum components on peptide stability. IC50 values obtained under the most stringent conditions were used to extrapolate the selectivity index (S.I.) toward salient bacterial species. In medium containing serum, all AMPs exhibited minimal to no cytotoxicity, with IC50 values exceeding 100 µg/mL. Dadapin-1 was the peptide that exhibited the lowest cytotoxicity, KSL-W exhibited the highest stability, and KSL exhibited the highest selectivity. Overall, these findings highlight the potential of these AMPs for the future production of anti-infective materials.
Keywords: AMPs; L929; MG63; antimicrobial peptides; cytocompatibility; cytotoxicity; hMSCs; implant orthopedic infections; proteolytic stability; selectivity index.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

