Alterations in the Rice Coleoptile Metabolome During Elongation Under Submergence Stress
- PMID: 39769021
- PMCID: PMC11678009
- DOI: 10.3390/ijms252413256
Alterations in the Rice Coleoptile Metabolome During Elongation Under Submergence Stress
Abstract
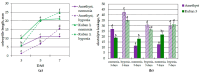
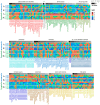
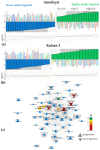
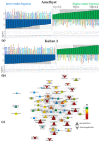
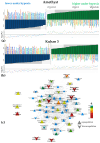
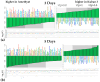
Plants known as obligate aerobes developed different mechanisms to overcome the damage incurred under oxygen limitation. One of the survival strategies to have commonly appeared in hydrophytic plants is the escape strategy, which accelerates plant axial organs' growth in order to escape hypoxic conditions as soon as possible. The present study aimed to distinguish the alterations in coleoptile elongation, viability and metabolic profiles in coleoptiles of slow- and fast-growing rice varieties. All the parameters were tested at 3, 5 and 7 days after sowing, to highlight changes during seedling development in normal and submerged conditions. The obtained results indicated that coleoptile elongation correlated with higher resistance to oxygen deprivation. GS-MS-based metabolic profiling indicated that coleoptiles of the fast-growing cultivar accumulated higher amounts of sugar phosphates, disaccharides, fatty acid derivatives and sterols, which are important for maintaining growth, membrane stability and viability. The slow-growing variety was characterized by a greater abundance of carboxylates, including lactate and phosphoric acid, indicating an energy crisis and cytosol acidification, leading to cell damage and low tolerance. Therefore, a metabolomics approach could be used for phenotyping (chemotyping) in the large-scale screening of newly developed varieties with higher tolerance to oxygen deprivation.
Keywords: Oryza sativa; adaptation; coleoptile; elongation; metabolomics; submergence; tolerance.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



Similar articles
-
Quantifying ATP turnover in anoxic coleoptiles of rice (Oryza sativa) demonstrates preferential allocation of energy to protein synthesis.J Exp Bot. 2012 Jul;63(12):4389-402. doi: 10.1093/jxb/ers114. Epub 2012 May 13. J Exp Bot. 2012. PMID: 22585748 Free PMC article.
-
Mechanisms of growth and patterns of gene expression in oxygen-deprived rice coleoptiles.Plant J. 2015 Apr;82(1):25-40. doi: 10.1111/tpj.12786. Plant J. 2015. PMID: 25650041
-
Identification of hub genes involved in gibberellin-regulated elongation of coleoptiles of rice seeds germinating under submerged conditions.J Exp Bot. 2024 Jul 10;75(13):3862-3876. doi: 10.1093/jxb/erae144. J Exp Bot. 2024. PMID: 38571323
-
Rice germination and seedling growth in the absence of oxygen.Ann Bot. 2009 Jan;103(2):181-96. doi: 10.1093/aob/mcn121. Epub 2008 Jul 25. Ann Bot. 2009. PMID: 18660495 Free PMC article. Review.
-
Metabolic adaptation to sugar/O2 deficiency for anaerobic germination and seedling growth in rice.Plant Cell Environ. 2014 Oct;37(10):2234-44. doi: 10.1111/pce.12311. Epub 2014 Apr 9. Plant Cell Environ. 2014. PMID: 24575721 Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

