The Astragalus Membranaceus Herb Attenuates Leukemia by Inhibiting the FLI1 Oncogene and Enhancing Anti-Tumor Immunity
- PMID: 39769192
- PMCID: PMC11676164
- DOI: 10.3390/ijms252413426
The Astragalus Membranaceus Herb Attenuates Leukemia by Inhibiting the FLI1 Oncogene and Enhancing Anti-Tumor Immunity
Abstract
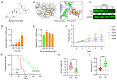
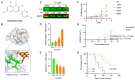
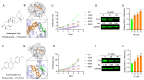
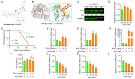
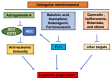
Astragalus membranaceus (AM) herb is a component of traditional Chinese medicine used to treat various cancers. Herein, we demonstrate a strong anti-leukemic effect of AM injected (Ai) into the mouse model of erythroleukemia induced by Friend virus. Chemical analysis combined with mass spectrometry of AM/Ai identified the compounds Betulinic acid, Kaempferol, Hederagenin, and formononetin, all major mediators of leukemia inhibition in culture and in vivo. Docking analysis demonstrated binding of these four compounds to FLI1, resulting in downregulation of its targets, induction of apoptosis, differentiation, and suppression of cell proliferation. Chemical composition analysis identified other compounds previously known having anti-tumor activity independent of the FLI1 blockade. Among these, Astragaloside-A (As-A) has marginal effect on cells in culture, but strongly inhibits leukemogenesis in vivo, likely through improvement of anti-tumor immunity. Indeed, both IDO1 and TDO2 were identified as targets of As-A, leading to suppression of tryptophane-mediated Kyn production and leukemia suppression. Moreover, As-A interacts with histamine decarboxylase (HDC), leading to suppression of anti-inflammatory genes TNF, IL1B/IL1A, TNFAIP3, and CXCR2, but not IL6. These results implicate HDC as a novel immune checkpoint mediator, induced in the tumor microenvironment to promote leukemia. Functional analysis of AM components may allow development of combination therapy with optimal anti-leukemia effect.
Keywords: Astragalus membranaceus; DTO2; FLI1; HDC; IDO1; drug–protein interaction; leukemia inhibition; traditional Chinese medicine; tumor microenvironment.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



Similar articles
-
Advancements in the Research of Astragalus membranaceus for the Treatment of Colorectal Cancer.Am J Chin Med. 2025;53(1):119-146. doi: 10.1142/S0192415X25500065. Epub 2025 Jan 29. Am J Chin Med. 2025. PMID: 39880662 Review.
-
FLI1 Regulates Histamine Decarboxylase Expression to Control Inflammation Signaling and Leukemia Progression.J Inflamm Res. 2023 May 10;16:2007-2020. doi: 10.2147/JIR.S401566. eCollection 2023. J Inflamm Res. 2023. PMID: 37193069 Free PMC article.
-
Research Progress on the Anti-Cancer Effects of Astragalus membranaceus Saponins and Their Mechanisms of Action.Molecules. 2024 Jul 18;29(14):3388. doi: 10.3390/molecules29143388. Molecules. 2024. PMID: 39064966 Free PMC article. Review.
-
Mechanism of Astragalus membranaceus (Huangqi, HQ) for treatment of heart failure based on network pharmacology and molecular docking.J Cell Mol Med. 2024 May;28(10):e18331. doi: 10.1111/jcmm.18331. J Cell Mol Med. 2024. PMID: 38780500 Free PMC article.
-
Astragaloside IV derived from Astragalus membranaceus: A research review on the pharmacological effects.Adv Pharmacol. 2020;87:89-112. doi: 10.1016/bs.apha.2019.08.002. Epub 2019 Dec 18. Adv Pharmacol. 2020. PMID: 32089240 Review.
References
MeSH terms
Substances
Grants and funding
- U1812403, 21867009 and 82260040/the Natural National Science Foundation of China
- QKHPTRC [2019]5627/he Science and Technology Department of Guizhou Province innovation and project grants
- QKHJC-ZK [2022]YB297, QKHJC-ZK[2023]YB240/the Science and Technology Department of Guizhou Province grants
- GZCNP202203Z/the Key Laboratory of Chemistry for Natural Products of Guizhou Province and Chinese Aca-demic of Sciences Research Grant
- RN21025/the Guizhou Medical University Research Grant
LinkOut - more resources
Full Text Sources
Research Materials

